by Jason Wasserman MD PhD FRCPC
November 23, 2025
Adenocarcinoma is the most common type of cancer that starts in the small intestine, the long, narrow part of the digestive system that absorbs nutrients from food. This type of cancer begins in the gland-forming cells that line the inside surface of the intestine. These cells normally help with digestion and nutrient absorption, but in adenocarcinoma, they grow in an abnormal, uncontrolled way. Small intestinal adenocarcinoma can occur anywhere along the small bowel, although it is most often seen in the duodenum, the segment closest to the stomach. Because the small intestine is located deep inside the abdomen and symptoms usually appear late, this cancer can be challenging to detect early. As it grows, it can spread into deeper layers of the intestinal wall, nearby organs, lymph nodes, and, in more advanced cases, other parts of the body.
Anatomy of the small intestine
The small intestine is a long tube that connects the stomach to the large intestine. It has three parts—the duodenum, jejunum, and ileum—which work together to break down food and absorb nutrients. The duodenum sits closest to the stomach and receives partially digested food mixed with stomach acid and bile. This area’s constant exposure to acid and digestive enzymes is thought to be one reason why adenocarcinomas are most common here. The jejunum and ileum make up the middle and final portions of the small intestine. Tumors can develop in these segments as well, but they are less common.
What are the symptoms of adenocarcinoma of the small intestine?
Adenocarcinoma of the small intestine usually does not cause symptoms in its early stages. When symptoms do occur, they depend on the size and location of the tumor. Many people experience abdominal pain, which may be constant or come and go. Some tumors bleed slowly, causing occult bleeding, which means small amounts of blood are present in the stool but not visible. Ongoing bleeding can lead to anemia, which may cause tiredness or shortness of breath.
As the tumor grows, it may block part of the intestine, causing nausea, vomiting, cramping, distension, or an inability to pass stool or gas. Tumors in the duodenum may also cause jaundice if they block the flow of bile. Some people experience unintentional weight loss, changes in appetite, or general fatigue. Because these symptoms are nonspecific, imaging or endoscopic procedures are often needed to identify the tumor.
What risk factors are associated with adenocarcinoma of the small intestine?
Several medical and lifestyle factors increase the risk of developing small intestine adenocarcinoma. Conditions that cause long-term inflammation, such as Crohn’s disease or celiac disease, increase risk by damaging the intestinal lining over time. Surgical procedures that expose the small intestine to unusual chemicals or digestive fluids, such as an ileal conduit, may also elevate the risk of cancer. Lifestyle factors such as smoking, heavy alcohol use, or a diet low in fruits and vegetables have been linked to a higher risk as well.
Some hereditary genetic syndromes increase the risk of small intestine cancer. These include familial adenomatous polyposis (FAP), which causes numerous polyps throughout the digestive tract; Lynch syndrome, which reduces the cell’s ability to repair DNA damage; and Peutz-Jeghers syndrome, which causes specific types of polyps in the small intestine. People with these conditions are often monitored closely for early signs of cancer.
Ampullary adenocarcinoma and non-ampullary adenocarcinoma
The duodenum contains a small but important structure called the ampulla of Vater, where the bile duct and pancreatic duct empty into the intestine. An adenocarcinoma that starts away from the ampulla is called non-ampullary adenocarcinoma, while a tumor that begins in or near the ampulla is called ampullary adenocarcinoma. These two types behave differently and often require different approaches to diagnosis and treatment.
Ampullary adenocarcinomas tend to cause symptoms earlier because even small tumors can block the flow of bile or pancreatic fluid. This may lead to jaundice, itching, dark urine, or light-colored stools. Non-ampullary adenocarcinomas, especially those located farther from the stomach, often grow silently until the tumor becomes large enough to cause bleeding or obstruction. Pathologists examine ampullary tumors slightly differently, and staging systems may differ from those used for non-ampullary small intestine adenocarcinoma. Despite these differences, both types are treated as serious cancers that benefit from careful pathological assessment and multidisciplinary care.
How is this diagnosis made?
Many people first undergo imaging tests such as CT or MRI scans when experiencing abdominal pain, anemia, or symptoms of intestinal blockage. These studies may show a thickened segment of intestine or a mass. Depending on the tumor’s location, an upper endoscopy or specialized endoscopic procedure may be performed to inspect the lining of the small intestine directly. In some cases, the entire small intestine is evaluated with capsule endoscopy, in which a tiny camera is swallowed to take thousands of images.
A biopsy is typically the first step toward a diagnosis. During a biopsy, a small piece of tissue is removed and examined under a microscope. In a biopsy, the pathologist looks for irregular glands, disorganized growth, enlarged nuclei, and other features suggesting adenocarcinoma. However, because biopsies contain only small samples, they may not show the full extent of the tumor.
A more complete diagnosis is usually made after the entire tumor is removed in a surgical procedure. The pathologist examines the tumor’s size, grade, depth of invasion, and whether it has spread into blood vessels, lymphatics, or nerves. The nearby lymph nodes are also evaluated for cancer. This information helps determine the cancer’s stage and guides treatment.
Immunohistochemistry is often performed to confirm the diagnosis or to rule out other tumour types that can occur in this area. Immunohistochemistry uses antibodies to detect proteins within cells. For example, specific staining patterns can distinguish adenocarcinoma from neuroendocrine tumours, lymphomas, or metastatic cancers from other organs.
Biomarkers for adenocarcinoma of the small intestine
Biomarkers are special tests performed on the tumor that help your doctors understand how the cancer behaves and whether certain treatments may be helpful. Two of the most important biomarker groups for adenocarcinoma of the small intestine are mismatch repair (MMR) proteins and HER2. These tests can be performed on biopsy tissue or on the tumor once it has been removed.
Mismatch repair proteins (MMR)
Mismatch repair testing looks for four proteins—MLH1, MSH2, MSH6, and PMS2—that normally help repair damage to DNA. When one or more of these proteins are lost, the tumor is called mismatch repair deficient (dMMR). Tumors with this pattern often show high levels of microsatellite instability (MSI), which is another way of measuring how well a cell can repair its DNA. Identifying a mismatch repair deficient tumor is important because these cancers may respond well to immune checkpoint inhibitors, a type of immunotherapy that can be used for many cancers regardless of where they start. Testing also helps identify patients who may have Lynch syndrome, an inherited condition that increases the risk of several types of cancer.
HER2
HER2 is another important biomarker. HER2 is a protein found on the surface of some cancer cells. Tumors with increased HER2 activity are called HER2-positive. Several targeted therapies exist for HER2-positive cancers, and although these therapies were originally developed for breast and stomach cancers, they are now being used more widely in advanced gastrointestinal cancers such as small intestine adenocarcinoma. If your tumor is advanced, has returned, or cannot be removed with surgery, your doctor may request HER2 testing to see if a targeted therapy might help.
Tests performed
Pathologists use immunohistochemistry, a test that highlights specific proteins in the tumor, to measure MMR protein levels and HER2 expression. In some cases, additional molecular tests such as PCR or next-generation sequencing may be used to detect MSI or other genetic changes. Because guidelines for HER2 testing vary between cancer types, many pathology reports will also state which scoring system or criteria were used. All of this information helps your healthcare team choose the most effective treatment options for your diagnosis.
Histologic grade
The grade describes how closely the cancer cells resemble normal small intestine cells when viewed under the microscope. Normal small intestine tissue contains glands, which are round or tube-shaped structures that help absorb nutrients. In adenocarcinoma, the cancer cells may still form glands, or they may lose this ability.
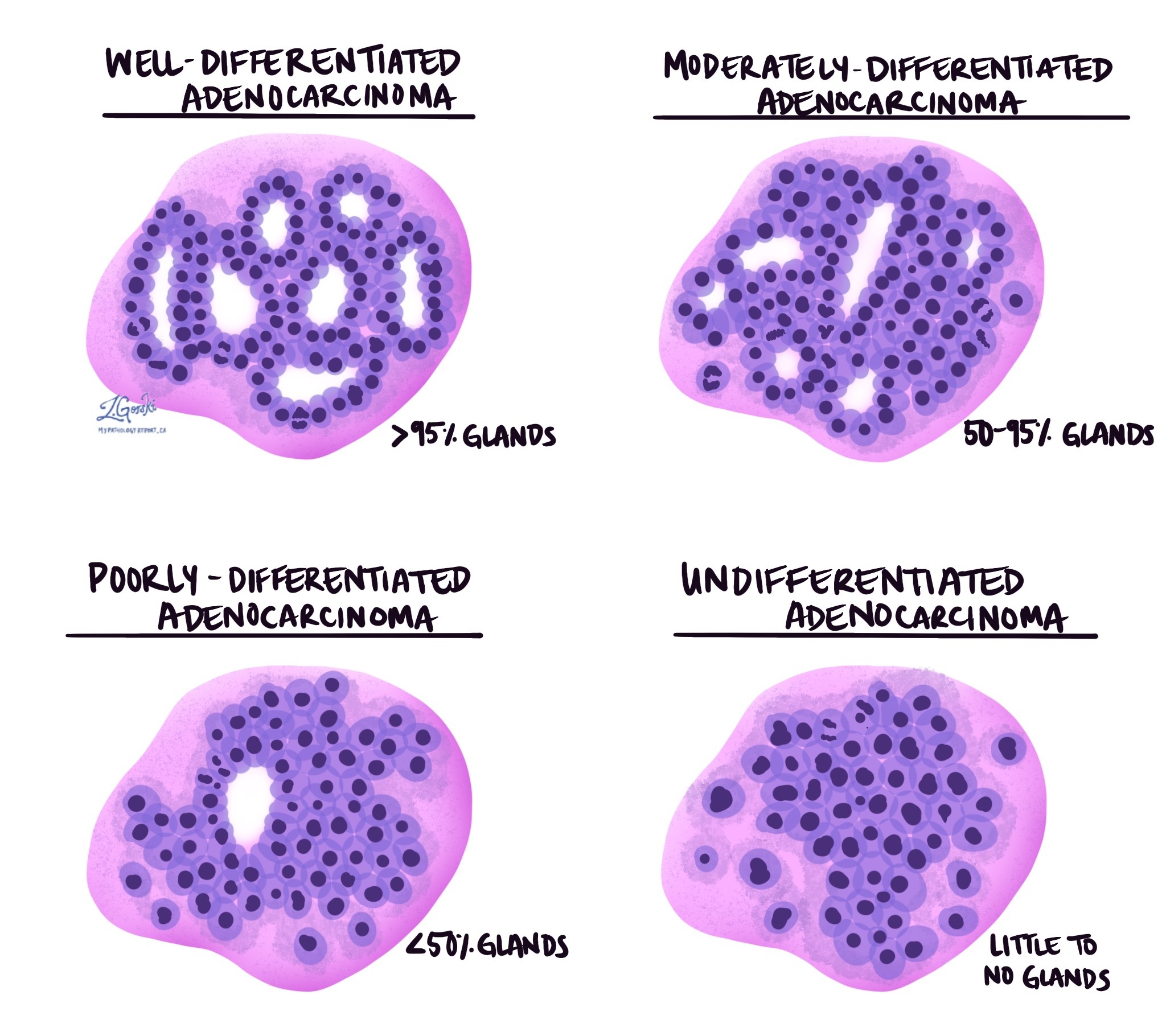
A tumor is described as well differentiated when the cancer cells form many glands and look somewhat like normal cells. Moderately differentiated tumors form fewer glands and show more variation in shape and size. Poorly differentiated tumors form very few recognizable glands and look much more abnormal. Some tumors are undifferentiated, meaning no glands can be identified at all. Higher-grade tumors tend to grow more quickly and are more likely to spread to lymph nodes or distant organs. Grade is an important factor in determining prognosis.
Tumor extension (depth of invasion)
Adenocarcinoma starts in the epithelium, the inner lining of the intestine. Beneath the epithelium is a thin layer of connective tissue called the lamina propria. When cancer cells invade the lamina propria, the diagnosis of adenocarcinoma can be made. Deeper to the lamina propria is the submucosa, a thick supportive layer containing blood vessels and lymphatic channels. Below that is the muscularis propria, a strong layer of muscle that moves food through the intestine. The outermost layer is the serosa, a thin protective covering.
As the tumor grows, it may move through each of these layers in order. Tumors that invade only the upper layers tend to behave less aggressively, while those that reach the muscularis propria, serosa, or nearby organs have a higher risk of spreading. The depth of invasion is a key part of the pathologic T stage and helps guide treatment decisions.
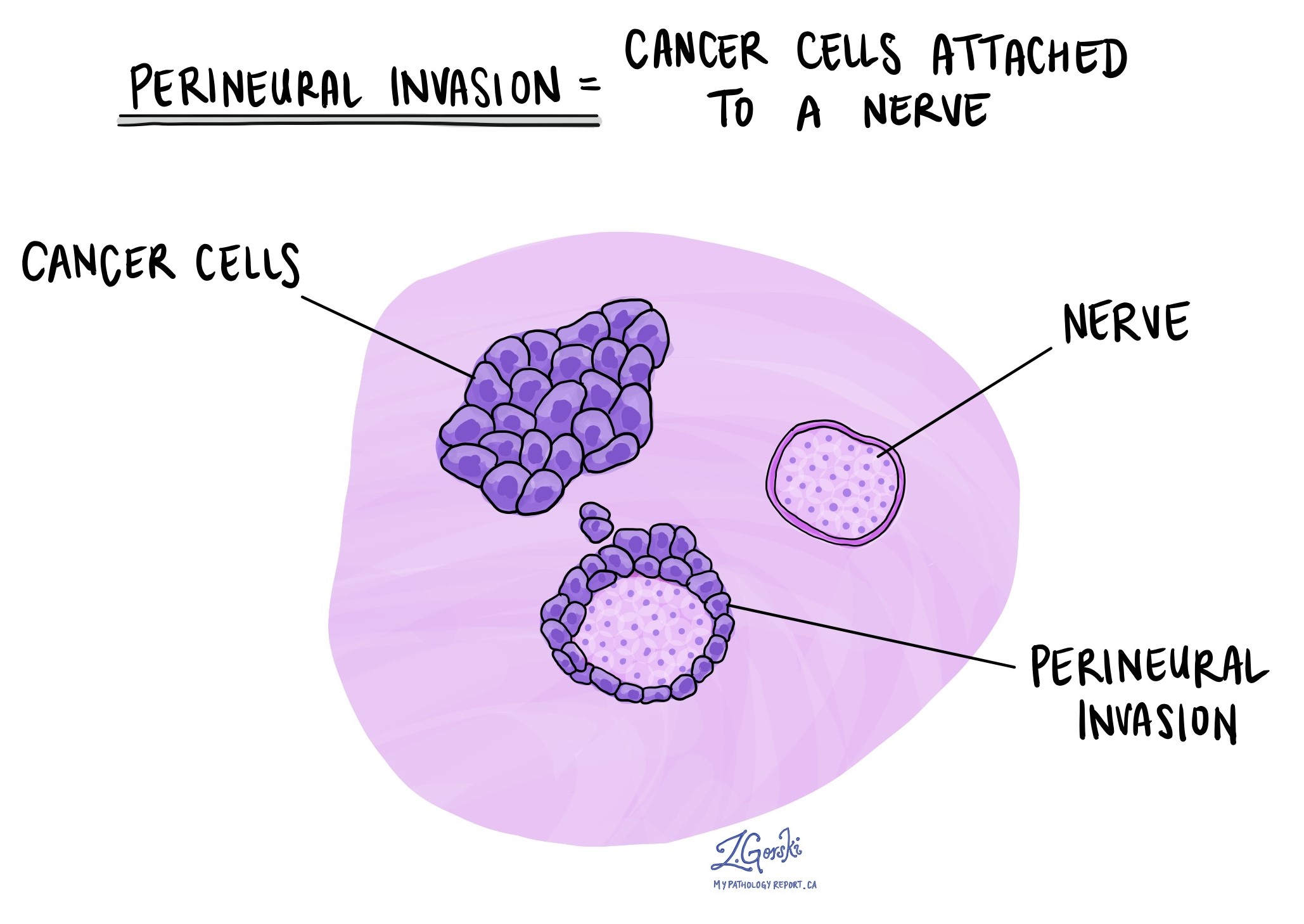
Perineural invasion
Perineural invasion means that cancer cells are seen around a nerve in the tissue surrounding the tumor. Nerves act like electrical cables, carrying signals such as pain or temperature between the body and the brain. When cancer cells grow along these nerves, they can travel deeper into surrounding tissues. Perineural invasion is important because its presence is associated with a higher chance that the tumor may come back after surgery, and it may also indicate a higher risk of spread.
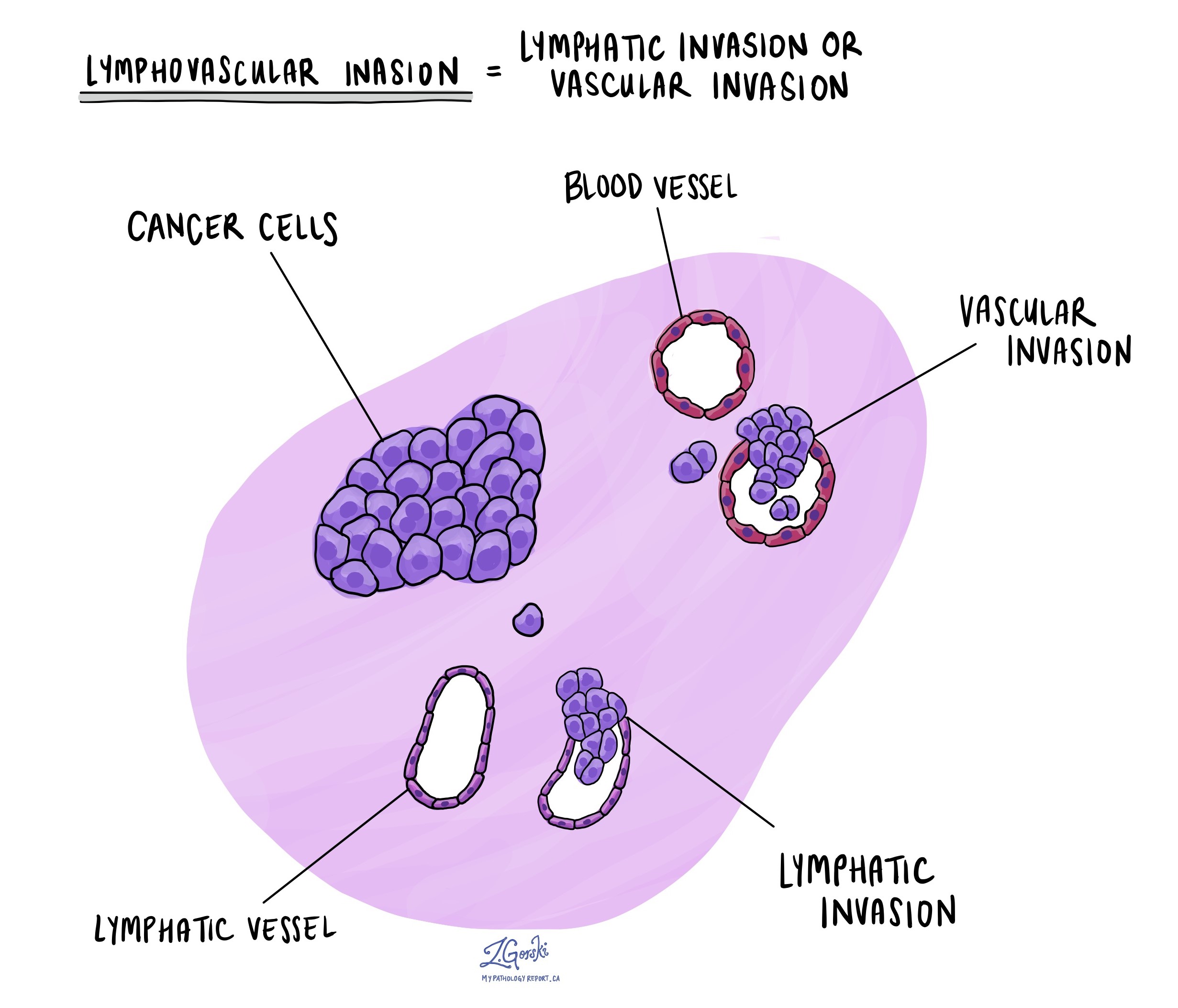
Lymphovascular invasion
Lymphovascular invasion occurs when cancer cells are found inside a blood vessel or lymphatic vessel near the tumor. Blood vessels carry blood throughout the body, while lymphatic vessels carry lymph fluid to lymph nodes, which are small immune organs. When cancer cells enter these vessels, they can travel to lymph nodes or distant organs such as the liver. Lymphovascular invasion is an important finding in the pathology report because it increases the likelihood of metastasis and affects the cancer’s stage and prognosis.
Margins
A margin is the edge of the tissue removed along with the tumour during surgery. The pathologist carefully examines the margins to determine whether the tumor was removed entirely. A negative margin means no cancer cells are seen at the cut edge, suggesting the tumor was entirely removed. A positive margin means cancer cells reach the cut edge, raising the possibility that some tumor remains in the body. When possible, the report will also describe the distance from the tumour to the nearest margin. Margin status is important because it helps guide decisions about further treatment.

Special microscopic features
Some adenocarcinomas have unique microscopic features that provide additional information about how the tumor may behave. Signet ring cell features occur when cells fill with mucus, pushing the nucleus to one side and giving the cell a ring-like appearance. These cells often do not stick together well, and tumors with this pattern may grow more aggressively. Mucinous features describe tumors that produce large amounts of mucus; if more than half of the tumor is made of mucus-producing cells, it is called a mucinous adenocarcinoma. Squamous differentiation is rare in small intestine adenocarcinoma, but it means that some cancer cells resemble squamous cells, the flat cells normally found in areas such as the skin. This feature may suggest a more aggressive form of the disease.
Lymph nodes
Lymph nodes near the tumor are typically removed during surgery and examined for cancer cells. Cancer can travel from the tumor into nearby lymphatic vessels and then into the lymph nodes, a process called metastasis. The pathologist will report the number of lymph nodes examined and the number that contain cancer. If cancer cells break through the outer capsule of the lymph node into the surrounding tissue, this is called extranodal extension. Lymph node involvement is an important part of the pathologic N stage and significantly influences prognosis and treatment planning.
Pathologic stage (pTNM)
The pathologic stage describes how far the cancer has spread. The stage is based on: how deeply the tumor has grown into the layers of the intestine (the T category), whether cancer has spread to nearby lymph nodes (the N category), and whether it has spread to distant organs (the M category). Tumours confined to the inner layers of the intestine are at a lower stage and generally have a better outlook than those that invade deeper layers or involve lymph nodes. Your healthcare team will combine the pathologic stage with imaging and clinical findings to determine the final stage. The stage plays a key role in selecting treatment and predicting prognosis.
T Categories:
- pT0: No tumour is detected in the small intestine.
- pTis: The tumour is limited to the inner lining, showing early cancerous changes (carcinoma in situ or high-grade dysplasia).
- pT1: The tumour has grown into the first layers beneath the lining:
- pT1a: Tumour is limited to the lamina propria, a thin layer of tissue just beneath the inner lining.
- pT1b: Tumour extends into the submucosa, a layer containing blood vessels and connective tissue.
- pT2: The tumour has grown into the muscularis propria, the thick muscle layer that helps move food.
- pT3: The tumour has penetrated through the muscularis propria into a layer called the subserosa or nearby connective tissues but has not broken through the outer layer (serosa).
- pT4: The tumour has either broken through the serosa (the outermost layer) or invaded nearby organs or structures, such as other parts of the small intestine, the pancreas, or bile ducts.
N Categories:
- pN0: No cancer is found in nearby lymph nodes.
- pN1: Cancer has spread to one or two nearby lymph nodes.
- pN2: Cancer has spread to three or more nearby lymph nodes.
Prognosis
The prognosis for adenocarcinoma of the small intestine depends on several factors, including the tumor’s stage, grade, and location. Tumors detected early, before they spread to lymph nodes or distant organs, have a better prognosis. High-grade tumors, deeper invasion into the intestinal wall, and evidence of lymphovascular or perineural invasion are associated with a higher risk of recurrence. Ampullary adenocarcinomas are often detected earlier than non-ampullary tumors because they block bile flow and cause noticeable symptoms, such as jaundice. As a result, ampullary tumors may have a slightly better prognosis when detected early. However, both types require careful evaluation and treatment planning.
After the diagnosis
Once the pathology report is completed, your doctor will review the results with you and discuss the next steps. Additional imaging studies may be performed to determine whether the cancer has spread. You may be referred to an oncologist, a doctor who specializes in cancer treatment, to discuss treatment options. Treatment often involves surgery, chemotherapy, or a combination of both. Your medical team will work with you to create a personalized care plan based on the tumor’s stage and your overall health. Follow-up care, including regular imaging and blood tests, may be recommended to monitor for recurrence.
Questions to ask your doctor
-
Where in the small intestine did my tumor start?
-
Was this an ampullary or non-ampullary tumor?
-
Did the tumor spread to lymph nodes or nearby organs?
-
What was the tumor’s grade, and what does that mean for my prognosis?
-
Were perineural or lymphovascular invasion seen in my pathology report?
-
Were the surgical margins clear?
-
Do I need additional imaging, treatment, or follow-up care?
-
What treatment options are available, and what are the potential benefits and risks?


