by Jason Wasserman MD PhD FRCPC
November 24, 2025
Ampullary adenocarcinoma is a type of cancer that starts in the ampulla of Vater, a small but important area where the bile duct and pancreatic duct join and empty into the first part of the small intestine (the duodenum). The inner lining of the ampulla is made up of gland-forming cells that help handle bile and pancreatic juices.
Because the ampulla is the main “exit point” for bile from the liver and enzymes from the pancreas, even a relatively small tumor in this area can block these ducts. This often causes symptoms such as jaundice and leads to earlier detection than cancers that start in other parts of the small intestine. Ampullary adenocarcinoma is considered a malignant tumor, meaning it can invade surrounding tissues and spread to lymph nodes and, in advanced cases, to distant organs.
Anatomy of the ampulla
The ampulla of Vater is located in the wall of the duodenum, just beyond the stomach. It is where the common bile duct (carrying bile from the liver and gallbladder) and the pancreatic duct (carrying digestive enzymes from the pancreas) join and open into the intestine through a small opening. For this reason, the ampulla is sometimes described as a small “junction box” that controls the flow of bile and pancreatic juices into the gut.
Ampullary adenocarcinomas arise from this small region and may grow predominantly toward the duodenal surface, within the ampullary cavity itself, or along the short ducts that lead into the ampulla. Distinguishing a true ampullary tumor from a cancer starting in the pancreas or bile duct is very important, and the pathologist and surgeon work together carefully to determine the tumor’s exact origin.
What are the symptoms of ampullary adenocarcinoma?
Ampullary adenocarcinoma often causes symptoms earlier than cancers in other parts of the small intestine because it can quickly block the bile duct. Many people develop jaundice, which is yellowing of the skin and eyes, along with dark urine, pale (clay-colored) stools, and itching. These symptoms happen because bile cannot drain properly and builds up in the body. Some patients experience abdominal pain, especially in the upper abdomen, and may notice nausea, vomiting, or discomfort after eating.
Tumors that grow more toward the duodenal side can cause bleeding into the intestine. This may lead to black or bloody stools or occult bleeding, which is blood that is not visible but causes anemia and fatigue. In some cases, blockage of the pancreatic duct can cause digestive problems or pancreatic insufficiency. Because many of these symptoms are noticeable and concerning, ampullary tumors are often found at an earlier stage than other small bowel cancers.
What risk factors are associated with ampullary adenocarcinoma?
For most people with ampullary adenocarcinoma, no clear cause is identified. However, a few factors are known to increase risk. Some hereditary conditions are associated with a higher chance of developing ampullary cancer, including familial adenomatous polyposis (FAP), Lynch syndrome, and Peutz–Jeghers syndrome. In these conditions, inherited changes in certain genes make it more likely for tumors to develop in the digestive tract, including the ampulla.
Chronic inflammatory conditions of the intestine, such as Crohn disease or celiac disease, which are important risk factors for other small bowel cancers, appear to play a smaller role in ampullary tumors. Lifestyle factors such as smoking, alcohol use, and diet may contribute to risk, but their exact impact on ampullary adenocarcinoma is less well defined than in other gastrointestinal cancers.
Ampullary vs non-ampullary adenocarcinoma
Although both ampullary and non-ampullary adenocarcinomas can occur near one another in the duodenum, they are considered distinct tumors. Ampullary adenocarcinomas start in the ampulla of Vater and almost always involve the opening where the bile and pancreatic ducts enter the intestine. Because of this location, they frequently cause bile duct obstruction and jaundice at an earlier stage.
Non-ampullary adenocarcinomas start away from the ampulla, on the duodenal wall or farther down the small intestine. These tumors may grow quite large before causing symptoms such as bleeding or obstruction. Pathologists use careful examination of the surgical specimen to determine whether the epicenter (center point) of the tumor is in the ampulla or elsewhere. This distinction is important because ampullary tumors have different staging rules, different microscopic patterns, and sometimes a different prognosis compared with non-ampullary tumors.
How is this diagnosis made?
Many patients with ampullary adenocarcinoma first have blood tests that show abnormal liver function, followed by imaging studies such as ultrasound, CT, or MRI that reveal blockage of the bile ducts. An endoscopic procedure (often an upper endoscopy or an ERCP, which combines endoscopy and X-ray imaging of the bile ducts) is then used to directly view the ampulla from inside the duodenum. The ampulla may appear enlarged, irregular, or ulcerated, and the doctor can take biopsies from this area.
The first diagnosis is usually made on the biopsy. Under the microscope, the pathologist looks for abnormal gland-forming cells, irregular architecture, and enlarged, crowded nuclei that confirm adenocarcinoma. Because the biopsy samples only a small portion of the lesion, it may not show every important feature, such as how deeply the tumor grows or whether it has spread into surrounding tissue.
A more complete evaluation is possible after surgery, when the entire tumor and surrounding tissues are removed, often in a procedure called a pancreaticoduodenectomy (Whipple procedure). The pathologist examines the tumor to confirm that it truly originates in the ampulla, measures its size, evaluates how deeply it has invaded, and checks nearby organs and tissues. The pathologist also assesses lymph nodes, blood vessels, lymphatic vessels, and nerves for signs of spread.
Immunohistochemistry is frequently used to help classify ampullary adenocarcinomas into intestinal type, pancreatobiliary type, or mixed type. This technique uses antibodies that bind to specific proteins in the cells, which can be seen under the microscope. Patterns of staining with markers such as CDX2, MUC1, MUC2, and MUC5AC can support one type or another. Although this classification can have prognostic and research value, the most important information for patients usually remains the tumor’s stage, grade, and whether it has spread.
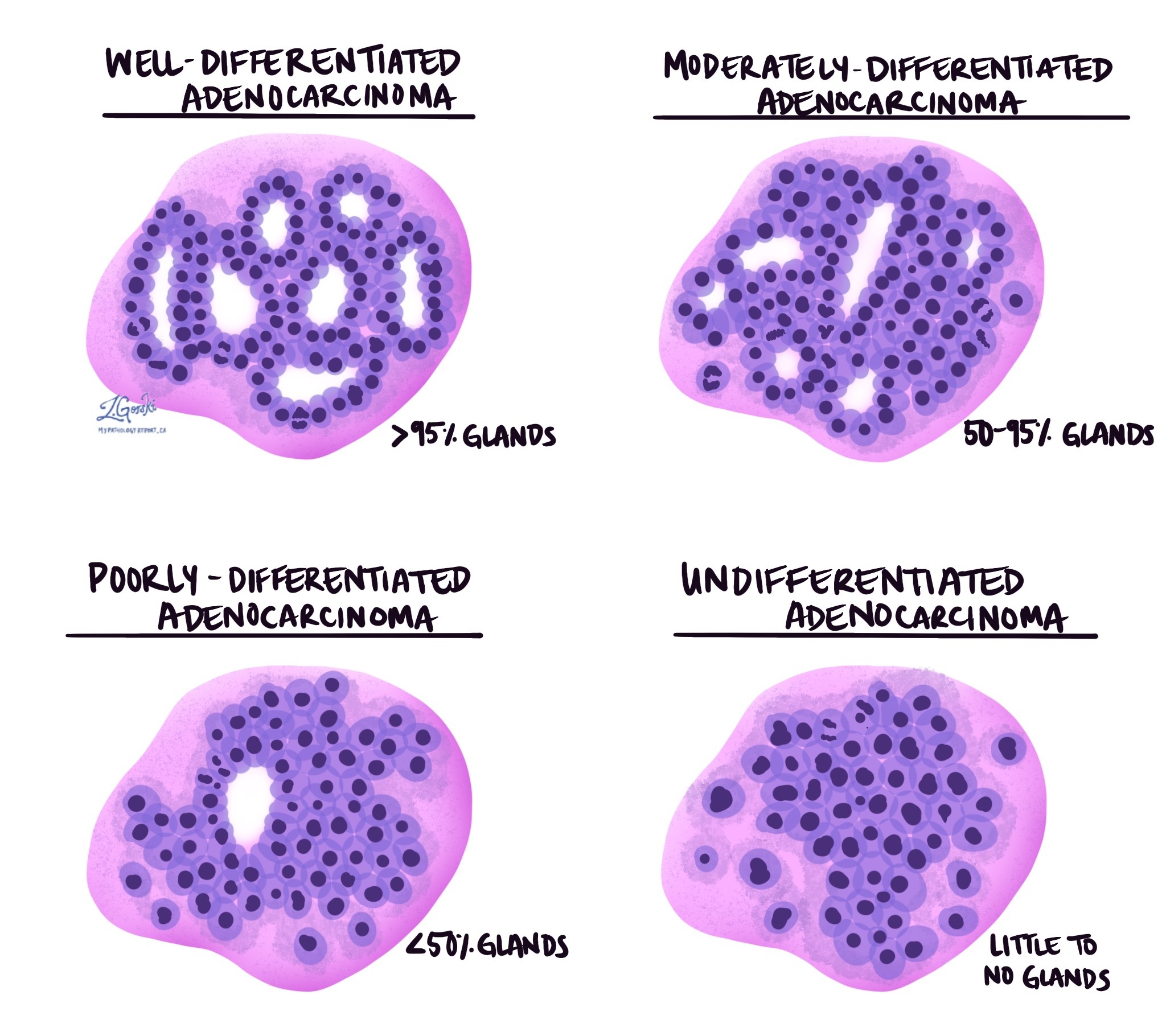
Histologic grade
The grade describes how closely the cancer cells resemble normal gland-forming cells of the ampulla and duodenum. Normal glands are round or tube-shaped structures that help move and process digestive fluids. In well-differentiated tumors, the cancer cells form many recognizable glands. Moderately differentiated tumors form fewer glands and show more variation in cell size and shape. Poorly differentiated tumors form very few glands and look more disorganized, and undifferentiated tumors lack gland formation altogether. Higher-grade tumors tend to grow more quickly and are more likely to spread, making grade an important factor in prognosis.
Histologic types
Ampullary adenocarcinomas can be divided into several histologic types, which describe how the tumor looks under the microscope and how the cancer cells are arranged. These types reflect the tumor’s cell of origin and sometimes help predict how the cancer will behave. In many reports, your pathologist will specify whether the tumor is intestinal type, pancreatobiliary type, mixed, or has one of several less common patterns. While histologic type is only one part of the overall picture, it can provide useful information about prognosis.
Intestinal type adenocarcinoma
Intestinal type adenocarcinoma looks under the microscope very similar to cancers that start in the colon. The tumor cells form glands (round or tube-shaped structures) lined by tall columnar cells, often with elongated, crowded nuclei. The pathologist may also see specialized cells such as goblet cells (which make mucus) or Paneth cells (normally found in the small intestine). These tumors often develop from an intestinal-type adenoma, which is a precancerous polyp-like growth.
In general, intestinal type ampullary adenocarcinomas tend to behave somewhat less aggressively than other types, especially when found at an early stage and completely removed. However, prognosis still depends mainly on stage, lymph node involvement, and other features such as lymphovascular and perineural invasion.
Pancreatobiliary type and gastric adenocarcinoma
Pancreatobiliary type adenocarcinoma resembles cancers that arise in the pancreas, gallbladder, or bile ducts. The glands are usually smaller and more widely separated by dense scar-like tissue called desmoplastic stroma. The cells tend to be cuboidal to low columnar and may look more irregular than in intestinal type tumors. Tumors with gastric-like (stomach-like) features are often grouped together with the pancreatobiliary type because they behave in similar ways.
Pancreatobiliary type ampullary adenocarcinomas are generally considered more aggressive than pure intestinal type tumors. They are more likely to invade deeply and spread to lymph nodes. Even so, they usually have a better prognosis than pancreatic ductal adenocarcinoma, which is important when discussing outcomes with patients.
Tubular adenocarcinoma with mixed features
Many ampullary adenocarcinomas show a mix of intestinal and pancreatobiliary features. In these tumors, some areas look like intestinal type, while others resemble pancreatobiliary type. When the pattern is mixed and cannot be clearly assigned to one group, the tumor is often reported as a tubular adenocarcinoma with mixed features, and the pathologist may comment on which pattern appears to predominate.
Because these tumors contain both components, their behavior tends to fall between that of pure intestinal and pure pancreatobiliary types. Prognosis is influenced more by stage and other high-risk features than by the exact proportion of each pattern. Still, noting mixed features helps give a more complete picture of the tumor’s biology.
Mucinous adenocarcinoma
Mucinous adenocarcinoma is a type in which a large portion of the tumor is made up of mucin, a jelly-like material produced by the cancer cells. Under the microscope, strips or clusters of tumor cells and glands appear to float in pools of mucin. These tumors often show an intestinal-like appearance and may be associated with adenomas in the ampullary region.
The impact of mucinous change on prognosis can vary. In some settings, mucinous tumors may behave more aggressively, especially if they are high grade or advanced stage. However, for ampullary cancers specifically, the most important factors remain depth of invasion, lymph node status, and margin status. The presence of mucinous features is one piece of information that helps your doctors understand the tumor, but it does not, by itself, determine the outcome.
Poorly cohesive cell carcinoma
Poorly cohesive cell carcinoma (sometimes including signet ring–like cells) is a rare type in which cancer cells do not stick together well. Instead of forming glands, the cells spread as single cells or small clusters through the tissue. These tumors are often diagnosed at a more advanced stage and tend to behave more aggressively than conventional gland-forming adenocarcinomas.
Because of their infiltrative growth pattern, poorly cohesive carcinomas can be more difficult to remove completely and are more likely to spread. Their presence usually indicates a higher-risk tumor, and this information is important when planning treatment and follow-up.
Medullary carcinoma
Medullary carcinoma is an uncommon subtype that can occur in the ampulla and duodenum. Under the microscope, these tumors form large, solid (sheet-like) masses with pushing borders and a prominent immune cell infiltrate. Despite their sometimes alarming appearance and size, medullary carcinomas often behave less aggressively than expected.
In many cases, medullary carcinomas are associated with mismatch repair deficiency (dMMR), which can have implications for immunotherapy. Overall, when compared with other high-grade tumors in this region, medullary carcinomas may have a more favorable prognosis, especially when detected early and completely removed.
Adenosquamous carcinoma
Adenosquamous carcinoma is a very rare type of ampullary cancer that contains both gland-forming (adenocarcinoma) areas and squamous areas. Squamous cells are flat cells similar to those found in the skin or lining of the esophagus. The pathologist confirms the squamous component with special stains and careful microscopic review.
Because adenosquamous carcinoma combines two aggressive patterns, it is generally considered a high-risk subtype with a tendency to behave more aggressively than typical tubular adenocarcinoma. However, its rarity makes it difficult to predict behavior in individual patients, so stage and other standard prognostic features remain crucial.
High-grade neuroendocrine carcinoma and mixed carcinomas
High-grade neuroendocrine carcinomas (NECs) are rare tumors in the ampulla that arise from neuroendocrine cells, which are specialized cells that release hormones and signaling molecules. These tumors can have a small-cell or large-cell pattern and typically grow rapidly. Some ampullary tumors contain both an adenocarcinoma component and a high-grade neuroendocrine component; these are often called mixed carcinomas.
High-grade NECs and mixed carcinomas are usually very aggressive, with a higher likelihood of early spread to lymph nodes and distant organs. They often require different treatment strategies, closer to those used for high-grade neuroendocrine cancers elsewhere in the digestive system. Their presence in a pathology report usually indicates a need for more intensive therapy and follow-up.
Undifferentiated carcinoma
Undifferentiated carcinoma is a term used when the tumor cells have lost all resemblance to normal glandular tissue and do not fit into the usual histologic categories. Some undifferentiated ampullary tumors contain giant cells or show a rhabdoid appearance (large cells with abundant pink cytoplasm and eccentric nuclei). These tumors often lack clear gland formation and may require additional immunohistochemical and molecular tests to classify.
Undifferentiated carcinomas of the ampulla are rare but highly aggressive. They tend to present at an advanced stage and have a higher risk of spread and recurrence. Because they are so uncommon, treatment decisions are often based on general principles used for other high-grade, poorly differentiated carcinomas, and prognosis is usually guarded.
Tumor extension (depth of invasion)
Ampullary adenocarcinoma begins in the ampulla of Vater, but as it grows, the cancer can move beyond this small structure into nearby tissues and organs. This process is called invasion, and it is one of the most important factors used to determine the pathologic tumor stage (pT). Pathologists can assess depth of invasion most accurately after the entire tumor has been removed during surgery. The deeper the tumor has grown into surrounding structures, the more advanced the cancer is and the greater the chance it may spread.
Tumors limited to the ampulla
In some cases, the tumor is still limited to the ampulla, the small opening where the bile duct and pancreatic duct meet the intestine. When a tumor is confined to the ampulla itself, it has not yet invaded the surrounding tissues. Tumors found at this early stage often present with symptoms such as jaundice, which leads to earlier detection. Because these cancers are still contained within a small, well-defined area, they generally have a more favorable prognosis.
Invasion of the sphincter of Oddi
As the tumor enlarges, it may grow beyond the sphincter of Oddi, a ring of muscle that surrounds the end of the bile duct and pancreatic duct and regulates the flow of digestive fluids into the duodenum. When the tumor moves past this muscular ring, it begins to infiltrate the surrounding soft tissue. Although invasion beyond the sphincter of Oddi indicates a more advanced tumor, the spread is usually still relatively localized at this stage.
Invasion of the duodenum
Ampullary adenocarcinoma may also extend into the duodenum, the first part of the small intestine. The earliest duodenal layer reached is the submucosa, a supportive layer beneath the surface lining that contains blood vessels, nerves, and connective tissue. Invasion into the submucosa means the tumor has breached the inner surface of the duodenum but has not yet reached the deeper muscular layer. Tumors at this stage show intermediate progression.
Invasion of muscularis propria
A deeper level of invasion occurs when the tumor reaches the muscularis propria, the thick band of muscle responsible for moving food through the intestine. Tumors that penetrate this muscle layer demonstrate more aggressive behavior and have an increased risk of reaching nearby organs or lymph nodes.
Invasion of the pancreas
Because the ampulla is located very close to the head of the pancreas, some ampullary adenocarcinomas grow directly into the pancreas. Invasion into the pancreas indicates a significant degree of spread and may affect pancreatic function. Tumors that extend into the pancreas are considered more advanced because treatment becomes more complex and the risk of recurrence increases.
Invasion of surrounding tissues
From the pancreas, the cancer can also extend into the peripancreatic soft tissue, which includes the fat and connective tissue surrounding the gland. Tumor invasion in this area typically indicates a high-stage tumor. These cancers can be more challenging to remove completely and are more likely to have already spread beyond the immediate region.
In some cases, the tumor may reach the serosa, the thin outer covering of the duodenum. This represents one of the most advanced levels of duodenal involvement. When the tumor breaks through the serosa, it is at the outermost boundary of the organ and has a greater potential to spread to neighboring structures.
Invasion of nearby organs
Very advanced tumors can invade adjacent organs. If the tumor grows upward, it may involve the stomach, which sits just above the duodenum. Spread toward the liver or bile ducts may lead to invasion of the gallbladder. In rare but serious cases, the tumor may grow into major blood vessels such as the superior mesenteric artery or celiac artery, which supply blood to the stomach, pancreas, and intestines. When a tumor invades major arteries or nearby organs, it represents a very advanced stage of disease. At this stage, surgery may be limited or not possible, and treatment decisions focus on controlling the disease and managing symptoms.
Understanding how far the tumor has spread helps your healthcare team determine the pathologic T stage, choose the most appropriate treatment, and provide information about prognosis. Depth of invasion is one of the most important factors used to guide care for ampullary adenocarcinoma.
Perineural invasion
Perineural invasion (PNI) means that cancer cells are found growing around nerves in the tissue near the tumor. Nerves are like small electrical cables that carry signals such as pain and temperature. When tumor cells surround or track along these nerves, they can move into deeper tissues and may be harder to completely remove with surgery. Perineural invasion is considered an adverse feature and is usually associated with a higher risk that the cancer might recur after treatment. This finding is routinely reported by pathologists because it helps guide follow-up and treatment decisions.
Lymphovascular invasion
Lymphovascular invasion (LVI) is present when cancer cells are seen inside a blood vessel or lymphatic vessel near the tumor. Blood vessels carry blood throughout the body, and lymphatic vessels carry lymph fluid to lymph nodes. Cancer cells that enter these vessels can travel to distant sites, including lymph nodes, liver, or lungs. Lymphovascular invasion therefore indicates a greater risk that the cancer has already spread or may spread in the future. This information is part of the overall assessment of stage and prognosis.
Margins
A margin is the edge of the tissue removed at surgery. After the tumor is removed, the pathologist examines all of the margins to see whether any cancer cells are present at the cut edge. A negative margin means no cancer cells are seen at the edge, suggesting the tumor has been completely removed. A positive margin means cancer cells are present at the cut surface, raising concern that some tumor may remain in the body.
In ampullary adenocarcinoma, several margins may be evaluated, including the pancreatic neck margin, bile duct margin, duodenal margin, and posterior soft tissue margins. When possible, the pathologist also measures the distance from the tumor to the closest margin, even when margins are negative. Margin status is an important factor in deciding whether additional treatments, such as chemotherapy or radiation, might be recommended.
Lymph nodes
Lymph nodes near the ampulla are usually removed during surgery so they can be examined for cancer. Cancer cells may spread from the ampulla into lymphatic vessels and then into regional lymph nodes. The pathologist records the number of lymph nodes examined and the number that contain metastases (cancer deposits). If cancer cells grow through the capsule of a lymph node into the surrounding tissue, this is called extranodal extension, and it is considered a higher-risk feature.
Lymph node involvement is a major part of the pathologic N stage. Tumors that have not spread to lymph nodes generally have a better prognosis than those with multiple positive nodes.
Pathologic stage (pTNM)
The pathologic stage combines information about the primary tumor (T), regional lymph nodes (N), and distant metastasis (M). For ampullary adenocarcinoma, staging follows a system that reflects the complex anatomy of this area. Tumors limited to the ampulla or with only minimal extension into nearby structures have a lower T stage. Tumors that extend into the pancreas, peripancreatic soft tissues, or surrounding organs are assigned a higher T stage.
The N stage is based on the number of lymph nodes with metastasis, and the M stage indicates whether the cancer has spread to distant organs such as the liver or lungs. Together, the T, N, and M categories are combined into an overall stage (for example, stage I, II, III, or IV). This overall stage is one of the most important pieces of information used to guide treatment and estimate prognosis.
Biomarkers
Biomarker testing helps doctors understand how the tumor behaves and whether specific treatments may be effective. In ampullary adenocarcinoma, two particularly important biomarker groups are mismatch repair (MMR) proteins and HER2, although other markers may be evaluated in selected cases.
MMR testing looks at four proteins—MLH1, MSH2, MSH6, and PMS2—that help repair DNA damage. If one or more of these proteins is missing, the tumor is called mismatch repair deficient (dMMR) and often shows microsatellite instability (MSI). Tumors with this profile may respond well to immune checkpoint inhibitors, a type of immunotherapy that can be used for several types of cancer. Identifying dMMR/MSI-high tumors can also suggest an inherited condition such as Lynch syndrome, so these results may have implications for family members as well.
HER2 is a protein found on the surface of some cancer cells. Tumors with increased HER2 expression or gene amplification are called HER2-positive. Several targeted therapies exist for HER2-positive cancers, and although these treatments were first developed for breast and stomach cancers, they are increasingly considered in advanced or metastatic ampullary and other gastrointestinal cancers.
Pathologists commonly use immunohistochemistry to assess MMR protein expression and HER2 status. In some cases, additional molecular tests such as PCR or next-generation sequencing may be used to identify MSI or other genetic changes. The pathology report may also state which scoring or interpretation criteria were used for HER2. These biomarker results help your healthcare team select the most appropriate therapy for your tumor.
Prognosis
The prognosis for ampullary adenocarcinoma is influenced by many factors, including stage, grade, lymph node status, depth of invasion, margin status, and the presence or absence of perineural and lymphovascular invasion. Overall, ampullary cancers generally have a better prognosis than pancreatic ductal adenocarcinomas but can still behave aggressively, particularly when diagnosed at a later stage.
Tumors that are detected early, before they have spread to lymph nodes or distant organs, are associated with higher long-term survival rates. Lymph node metastasis, positive margins, perineural and lymphovascular invasion, and advanced T stage are all features associated with a higher risk of recurrence. Molecular features and biomarker results, such as mismatch repair status or specific gene mutations, may also provide additional prognostic information in some cases.
After the diagnosis
After your pathology report confirms ampullary adenocarcinoma, your healthcare team will review the findings with you and discuss next steps. Additional imaging studies may be ordered to look for any spread of the disease. Many patients are referred to a multidisciplinary team, which may include a surgeon, medical oncologist, gastroenterologist, and sometimes a radiation oncologist.
Treatment often involves surgery, usually a pancreaticoduodenectomy, if the tumor can be removed and the patient is medically fit for a major operation. Depending on the stage and other risk factors, chemotherapy, immunotherapy, or targeted therapy may be recommended after surgery. For tumors that cannot be completely removed, treatment focuses on controlling the disease, relieving symptoms (such as bile duct obstruction), and maintaining quality of life. Regular follow-up visits, imaging, and blood tests are usually recommended to monitor for recurrence.
Questions to ask your doctor
- What type and grade of ampullary adenocarcinoma do I have (intestinal, pancreatobiliary, or mixed)?
-
How far has the tumor grown, and has it spread to lymph nodes or other organs?
-
Were perineural or lymphovascular invasion seen in my pathology report?
-
Were the surgical margins clear?
-
Have biomarker tests such as MMR and HER2 been performed on my tumor, and what do the results mean?
-
What treatment options do you recommend, and what are the potential benefits and risks?
-
What is the expected prognosis in my situation?
-
How will I be monitored after treatment?


