Jason Wasserman MD PhD FRCPC
November 28, 2025
Ductal adenocarcinoma is the most common type of pancreatic cancer. It starts from the cells that line the small tubes (ducts) inside the pancreas. These cells normally help transport digestive fluids made by the pancreas into the small intestine. In ductal adenocarcinoma, these cells begin to grow abnormally and invade the surrounding tissue. Over time, the cancer may spread to nearby organs or distant parts of the body.
Where in the pancreas is this tumour usually found?
About two-thirds of ductal adenocarcinomas are found in the head of the pancreas (the part closest to the small intestine). The rest are found in the body or tail of the gland. Most tumours are solitary, meaning there is only one tumour in the pancreas.
What are the symptoms of ductal adenocarcinoma?
Many people with ductal adenocarcinoma experience symptoms such as fatigue, loss of appetite, indigestion, weight loss, or pain in the upper abdomen or back. If the tumour blocks the bile duct, it can cause jaundice, which makes the skin and eyes look yellow. Some people are diagnosed after developing new-onset diabetes or depression. In more advanced cases, symptoms may be caused by the cancer spreading to the liver or other organs.
What causes ductal adenocarcinoma?
The most well-known risk factor is smoking. Other risk factors include obesity, diabetes, chronic pancreatitis, and a family history of pancreatic cancer. Heavy alcohol use may also increase risk. Some people inherit gene mutations (changes) that increase their risk of developing pancreatic cancer, including changes in BRCA2, CDKN2A, or other DNA repair genes.
How is this diagnosis made?
Ductal adenocarcinoma of the pancreas is usually diagnosed after a biopsy, where a small piece of tissue is removed and examined under the microscope by a pathologist. Most biopsies are performed using a fine needle during an endoscopic ultrasound (EUS), although some are taken during surgery. After confirming the diagnosis on a small biopsy sample, surgery may later be performed to remove the entire tumour if appropriate.
Microscopic features
Under the microscope, ductal adenocarcinoma is made up of abnormal glands that grow in a disorganized way and invade the surrounding pancreatic tissue. These glands are often irregular in shape and size and may contain mucin, a jelly-like substance commonly produced by pancreatic duct cells. The tissue surrounding the tumour usually shows a dense, fibrous reaction, which is a response to the invasive growth. In more aggressive tumours, the cancer cells may lose their gland-forming structure and instead form solid sheets or show marked variation in size and shape.
Immunohistochemistry
Pathologists often perform immunohistochemistry (IHC) to support the diagnosis. Immunohistochemistry is a test that uses antibodies linked to dyes to highlight specific proteins inside tumour cells. These staining patterns help confirm that the tumour started in the pancreas and rule out other types of cancer that may look similar under the microscope. Although no single protein identifies pancreatic ductal adenocarcinoma on its own, certain combinations of markers support the diagnosis. Most tumours produce proteins typically seen in pancreatic duct cells, such as CK7, CK8, CK18, and CK19, and may also show staining for CEA, CA19-9, CA125, MUC1 (EMA), MUC5AC, and DUPAN-2. These proteins are helpful not only for diagnosis but also because some are used to monitor response to treatment.
Other stains are negative, which helps exclude similar-appearing tumours. For example, ductal adenocarcinoma usually does not express vimentin, neuroendocrine markers such as synaptophysin or chromogranin A, or acinar markers such as trypsin or BCL10. This pattern helps distinguish ductal adenocarcinoma from neuroendocrine tumours or acinar cell carcinomas.
Many ductal adenocarcinomas also show loss of tumour-suppressor proteins such as SMAD4 (DPC4) and p16 (CDKN2A), which normally help control cell growth. Loss of these proteins supports a diagnosis of cancer. Another commonly altered protein is p53. In about 75 to 80 percent of cases, p53 is either overproduced or completely lost in tumour cells—both patterns are abnormal and support the diagnosis.
In challenging cases—such as tumours with unusual features or when it is unclear whether the cancer began in the pancreas or in a nearby organ—immunohistochemistry is especially important. The pathologist combines these test results with clinical information and imaging studies to make the final diagnosis.
Histologic grade
Histologic grade is a way for pathologists to describe how different the tumour cells look and behave compared to normal cells in the pancreas. The grade provides important information about how aggressive the tumour may be and helps guide treatment decisions.
To determine the grade, the pathologist examines the tumour under the microscope and looks at several key features:
-
Glandular differentiation: This refers to how well the tumour cells form gland-like structures. Tumours that closely resemble the normal glandular structure of the pancreas are considered well differentiated. Tumours that form fewer or disorganized glands are classified as moderately or poorly differentiated.
-
Mucin production: Mucin is a jelly-like substance normally made by glands in the pancreas. Tumour cells that produce less or irregular mucin may behave differently than those that produce more mucin.
-
Mitotic activity: Mitosis is the process by which cells divide. Pathologists count how many tumour cells are actively dividing. Tumours with high mitotic activity are more likely to grow and spread quickly.
-
Nuclear features: The nucleus is the control center of the cell. In cancer, tumour cells often have enlarged, irregular, or darkly stained nuclei. The degree of nuclear change contributes to the tumour grade.
If a tumour shows areas with different grades (called intratumoral heterogeneity), the highest grade is used for the final diagnosis. This approach ensures that even small areas of more aggressive tumour are taken into account.
Based on these features, ductal adenocarcinoma of the pancreas is grouped into three grades:
-
Well differentiated: The tumour cells look similar to normal cells, form organized glands, and divide slowly. These tumours usually grow and spread at a slower rate.
-
Moderately differentiated: The tumour cells show more differences from normal cells and divide more frequently. The gland formation is less organized.
-
Poorly differentiated: The tumour cells look very abnormal, often form few or no glands, and divide rapidly. These tumours are more aggressive and more likely to spread.
Tumour grade is an important factor in predicting a person’s outcome. Higher-grade tumours are linked to worse survival and may require more intensive treatment.
Histologic subtypes of ductal adenocarcinoma
Histologic subtypes are different patterns that cancer cells can form when examined under a microscope. These subtypes are determined by the shape, structure, and behavior of the tumor cells. Recognizing the subtype is important because some subtypes behave more aggressively, respond differently to treatment, or have a better or worse prognosis than others.
The following sections describe the histologic subtypes of ductal adenocarcinoma of the pancreas.
Adenosquamous carcinoma and squamous cell carcinoma
Adenosquamous carcinoma contains two types of cancer cells: gland-forming (adenocarcinoma) and squamous cells. Squamous cells are not normally found in the pancreas and are usually associated with skin and mucosal surfaces. In this subtype, at least 30 percent of the tumor is made up of squamous cells. Under the microscope, these cells appear as sheets of polygonal cells with sharp borders and dense pink cytoplasm.
This subtype tends to be more aggressive than typical ductal adenocarcinoma. Pure squamous cell carcinoma of the pancreas is extremely rare, and in such cases, doctors must make sure the cancer has not spread from another part of the body such as the lungs.
Colloid carcinoma
Colloid carcinoma is a rare subtype in which the tumor cells float in large pools of mucin, a thick, jelly-like substance normally produced by some types of cells. Under the microscope, the cancer cells appear to be suspended in mucin rather than forming tightly packed glands.
This subtype is often linked to a non-invasive tumor called an intestinal-type intraductal papillary mucinous neoplasm (IPMN). Colloid carcinoma has a better prognosis than most other pancreatic cancers, with a higher five-year survival rate.
Hepatoid carcinoma
Hepatoid carcinoma is a very rare type of pancreatic cancer that looks like liver cancer under the microscope. The tumor cells are large and have pink cytoplasm similar to liver cells. They may also produce a protein called alpha-fetoprotein (AFP), which is normally made by the liver.
Because many tumors can mimic liver cancer, this diagnosis requires special tests to confirm the origin. In some cases, hepatoid carcinomas may behave aggressively, but there is limited information on prognosis due to how rare it is.
Medullary carcinoma
Medullary carcinoma is a rare subtype made up of sheets or nests of poorly differentiated tumor cells that grow in a pushing rather than infiltrating pattern. These tumors often have a strong immune response, with many immune cells found within and around the tumor.
Although medullary carcinomas are poorly differentiated, some patients with this type of tumor may have a better outcome than expected. This subtype is often associated with microsatellite instability and may respond to immunotherapy.
Invasive micropapillary carcinoma
In this subtype, at least half of the tumor is made up of small groups of cancer cells surrounded by clear spaces. These groups of cells appear to float within the tissue, forming what pathologists describe as a micropapillary pattern.
Micropapillary carcinomas tend to be more aggressive and may spread more easily to lymph nodes and other areas of the body.
Signet-ring cell carcinoma
Signet-ring cell carcinoma is an extremely rare form of pancreatic cancer. The tumor is made up of individual cells or small groups of cells that contain mucin, which pushes the nucleus to the side, giving the cell a “signet-ring” appearance under the microscope.
This type of cancer is often associated with tumors from other parts of the body, such as the stomach or breast, so doctors must carefully rule out a metastasis before diagnosing it as a primary pancreatic tumor. Signet-ring cell carcinomas tend to behave aggressively.
Undifferentiated carcinoma
Undifferentiated carcinoma is a high-grade cancer in which the tumor cells no longer resemble normal cells and do not show a clear pattern of differentiation. These tumors often grow as solid sheets of cells and may lack gland formation.
There are several patterns of undifferentiated carcinoma:
-
Anaplastic undifferentiated carcinoma contains highly abnormal cells with large, irregular nuclei and sometimes bizarre-looking giant cells. These tumors are often very aggressive.
-
Sarcomatoid undifferentiated carcinoma includes spindle-shaped tumor cells that may look like bone or cartilage. In some cases, the tumor contains rhabdoid cells, which are large and poorly cohesive.
-
Carcinosarcoma is a rare tumor that contains both glandular (epithelial) and spindle (sarcomatoid) components. Each component must make up at least 30 percent of the tumor.
Undifferentiated carcinomas generally have a poor prognosis and tend to grow and spread quickly.
Undifferentiated carcinoma with osteoclast-like giant cells
This rare tumor contains three types of cells. The first are non-cancerous osteoclast-like giant cells, which are large cells with many nuclei. The second are mononuclear histiocytic cells, which are part of the body’s immune system. The third type are the cancer cells, which can vary in shape and size.
Despite the presence of highly abnormal cancer cells, some tumors in this group behave less aggressively, and many patients live for years after diagnosis. This subtype is linked to typical ductal adenocarcinoma and shares some of the same genetic changes.
Tumour extension and pathologic tumor stage (pT)
Ductal adenocarcinoma of the pancreas starts in small channels called ducts that carry digestive enzymes produced by the pancreas. These ducts are found within the pancreas and are lined by a thin layer of specialized epithelial cells. As the tumour grows, it can move beyond the ducts and invade deeper layers of the pancreas and nearby tissues, including nerves, blood vessels, fat, and organs around the pancreas.
The extent of tumour growth, especially how far the tumour has spread within and beyond the pancreas, is one of the most important factors in understanding how advanced the cancer is and predicting a person’s outcome. This information is used to assign a pathologic tumor stage, often shortened to pT.
Pathologists determine the pT stage by measuring the size of the invasive tumour and identifying any spread into critical nearby structures such as major arteries. The pT stage is part of the TNM staging system used to describe cancer. A higher pT stage usually means the tumour is larger or has invaded important nearby structures, and it may be more difficult to remove with surgery.
Below is a simplified explanation of the pT stages used for ductal adenocarcinoma of the pancreas:
- pT0: No tumour was found.
-
pTis (carcinoma in situ): The tumour cells are present only within the ducts and have not invaded the surrounding pancreatic tissue. This includes high-grade precancerous changes such as PanIN-3, intraductal papillary mucinous neoplasms with high-grade dysplasia, and similar lesions.
-
pT1: The tumour is 2 cm or smaller.
-
pT1a: Tumour is 0.5 cm or smaller.
-
pT1b: Tumour is between 0.5 cm and 1 cm.
-
pT1c: Tumour is between 1 and 2 cm.
-
-
pT2: The tumour is larger than 2 cm but not more than 4 cm.
-
pT3: The tumour is larger than 4 cm but has not grown into nearby major arteries.
-
pT4: The tumour has grown into one or more major arteries such as the celiac axis, superior mesenteric artery, or common hepatic artery. The size of the tumour does not matter at this stage.
Understanding the pT stage is important because it helps guide treatment decisions and provides information about prognosis. Lower-stage tumours are often easier to remove completely and are associated with better outcomes. Higher-stage tumours may require more complex treatment and are more likely to recur or spread to other parts of the body.
Margins
Margins refer to the edges or borders of tissue that are cut during surgery to remove a tumour. After the surgery, a pathologist carefully examines these margins under the microscope to check if any cancer cells are present at the very edge of the removed tissue.
-
A negative margin (also called a clear margin) means no cancer cells are seen at the edge. This suggests that the tumour was likely removed completely.
-
A positive margin means cancer cells are seen right at the edge of the tissue. This raises concern that some cancer may have been left behind.
The margin status is important because it helps doctors decide whether additional treatment is needed. For example, if a margin is positive, further surgery or radiation therapy may be recommended to reduce the risk of the cancer coming back.
In pancreatic surgery, several specific margins are commonly evaluated:
-
Pancreatic transection margin: This is the edge of the pancreas that was cut to remove the tumour. It is often the most important margin when the tumour is located in the head or neck of the pancreas.
-
Common bile duct margin: This is the edge of the bile duct that was removed with the pancreas. The bile duct carries bile from the liver to the intestine and passes through or near the head of the pancreas.
-
Uncinate (retroperitoneal) margin: This is the deep tissue margin located behind the pancreas. It is close to important blood vessels and nerves and is frequently examined for tumour involvement.
-
Duodenal and gastric margins: If part of the small intestine (duodenum) or stomach is removed during surgery, the cut edges of these organs are also examined.
A report may also include the distance between the tumour and the closest margin. Even if a margin is not positive, a very close margin (such as less than 1 mm) may still increase the risk of recurrence.
Knowing whether the margins are negative or positive is essential for planning the next steps in care and estimating the likelihood of the cancer returning.
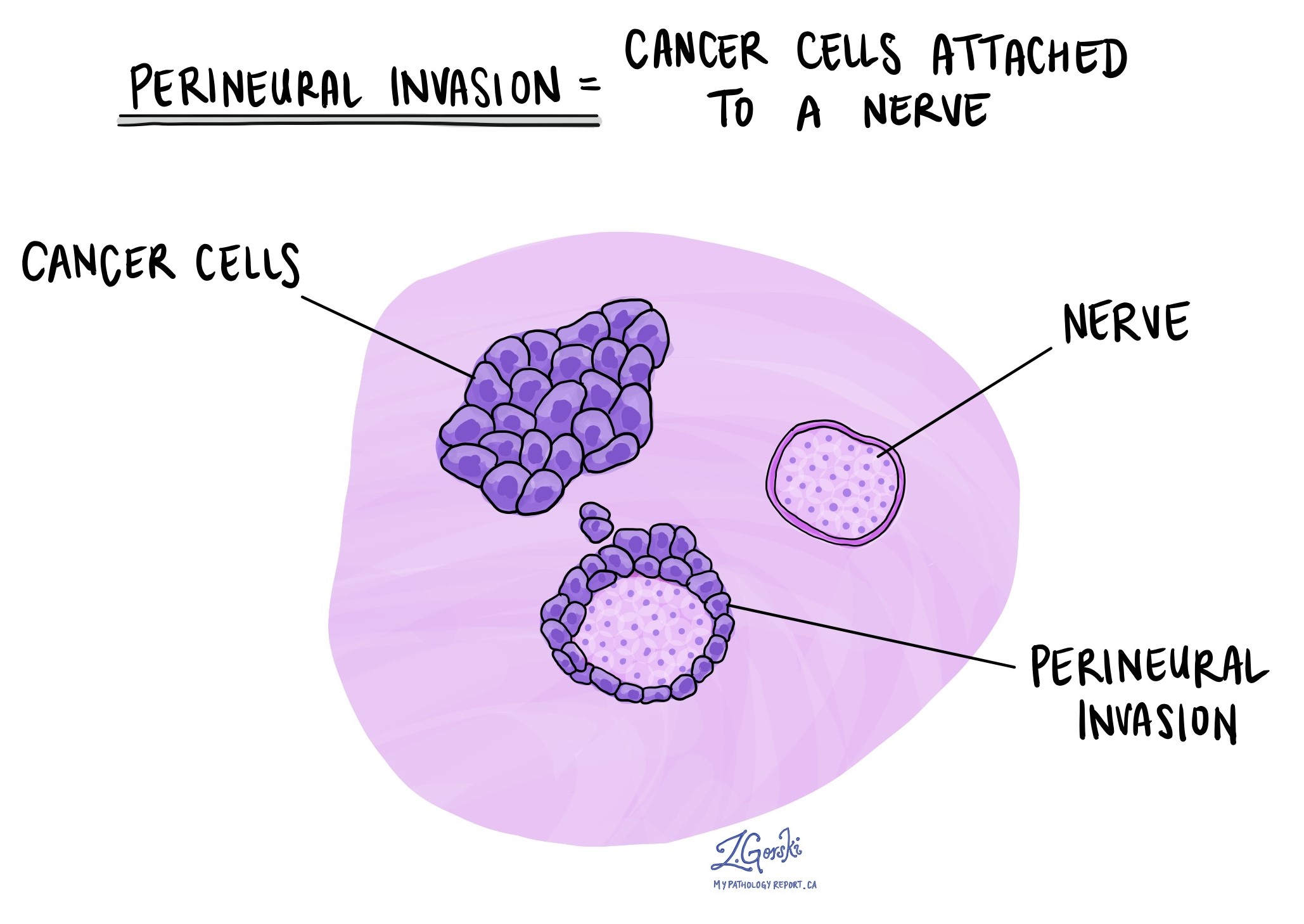
Perineural invasion
Perineural invasion (PNI) means cancer cells are growing along or around nerves. This is an aggressive feature that may increase the chance of cancer spreading. If present, it will be described in your pathology report.
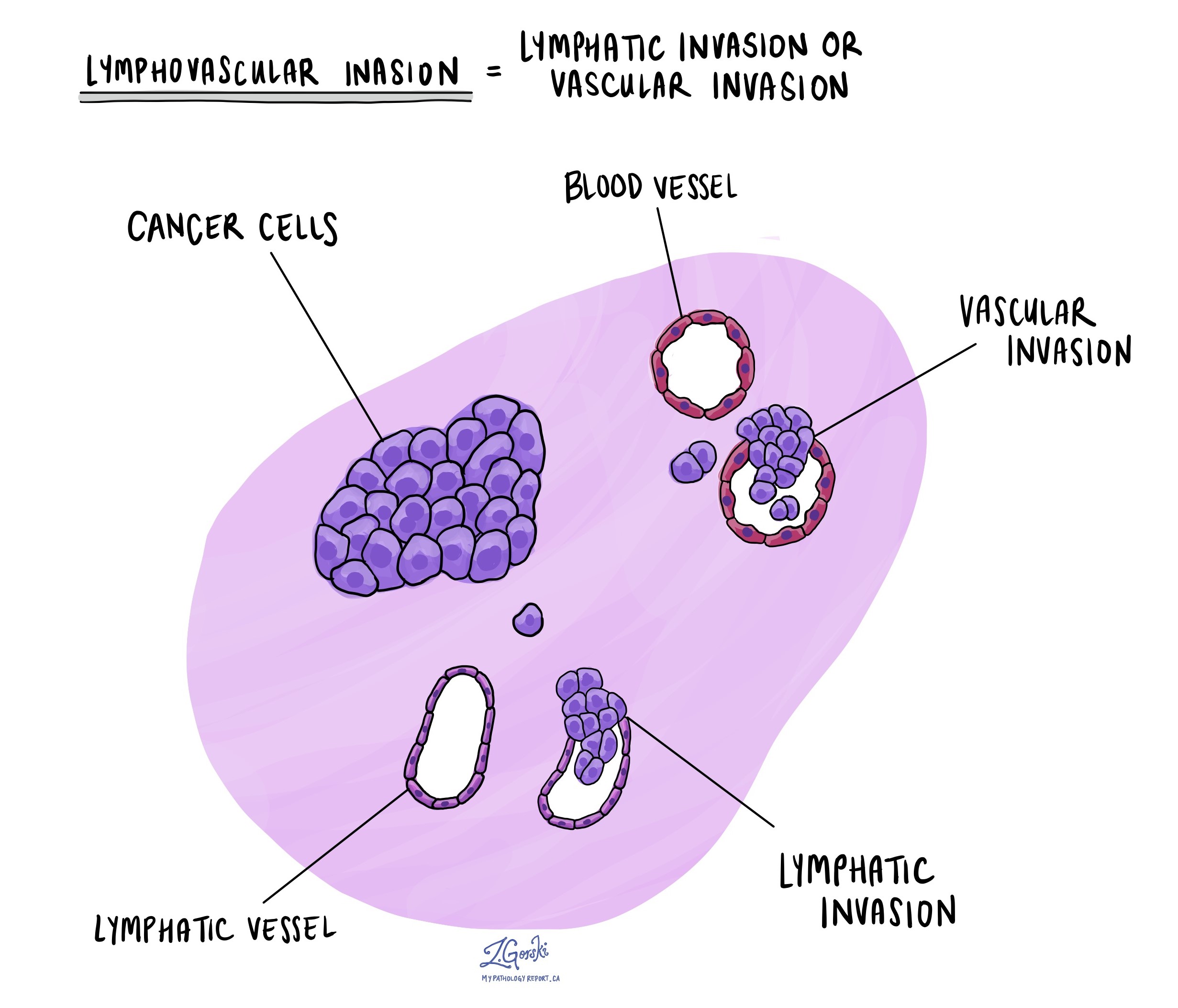
Lymphovascular invasion
Lymphovascular invasion (LVI) means that cancer cells are seen inside blood vessels or lymphatic vessels near the tumor. This increases the risk that cancer may spread to lymph nodes or other parts of the body.
Lymph nodes
Lymph nodes are small, bean-shaped organs that are part of the immune system. They help filter harmful substances from the body and are often the first place cancer spreads from the pancreas. During surgery for ductal adenocarcinoma of the pancreas, nearby lymph nodes are typically removed and examined under the microscope by a pathologist.
In your pathology report, the lymph nodes may be described in several ways:
-
The total number of lymph nodes examined.
-
The number of nodes containing cancer (also called positive nodes).
-
The location of the affected lymph nodes.
-
The size of the largest area of cancer in the nodes.
-
Whether the cancer has spread outside the lymph node into the surrounding tissue.
This information is used to determine the pathologic nodal stage (pN), which helps doctors understand how far the cancer has spread and guides decisions about treatment.
Pathologic nodal stages
-
pN not assigned: No lymph nodes were submitted or found in the tissue removed during surgery.
-
pN not assigned (cannot be determined): Lymph nodes were submitted but could not be properly assessed due to technical reasons or incomplete information.
-
pN0: No cancer was found in any of the lymph nodes examined.
-
pN1: Cancer was found in one to three regional lymph nodes.
-
pN2: Cancer was found in four or more regional lymph nodes.
The number of positive lymph nodes is important because it is strongly linked to prognosis. People with more lymph nodes containing cancer are more likely to have the disease come back after surgery. This information is also used when determining the overall stage of the cancer and whether additional treatment, such as chemotherapy, is recommended.
Biomarkers for adenocarcinoma of the pancreas
Biomarkers are measurable changes inside tumour cells—usually involving specific genes or proteins—that help doctors understand how the cancer behaves and which treatments may be most effective. Biomarker testing is especially important in pancreatic ductal adenocarcinoma because some tumours carry inherited or acquired genetic alterations that influence prognosis, guide the use of targeted therapy, and can identify family members who may also be at increased risk of cancer. Biomarker results help your healthcare team choose treatments such as PARP inhibitors, immunotherapy, or medications directed at specific gene mutations.
What types of biomarkers are tested in pancreatic cancer?
Most biomarker testing in pancreatic cancer examines the tumour’s DNA, looking for inherited or tumour-specific mutations that affect growth pathways or DNA repair systems. These tests may detect mutations in genes such as BRCA1, BRCA2, PALB2, and KRAS, or identify gene fusions such as RET. Additional tests may evaluate genes involved in mismatch repair (MMR), including MLH1, MSH2, MSH6, and PMS2, to determine whether immunotherapy may be helpful. Testing is typically performed using next-generation sequencing (NGS) on a biopsy or surgical specimen.
BRAF
BRAF is a gene involved in a growth pathway that helps regulate how cells divide. Certain mutations, particularly the V600E mutation, can make cancer cells grow more quickly. BRAF is important because tumours with BRAF mutations may respond to BRAF-targeted therapies, which block the abnormal protein and slow tumour growth.
Pathologists test for BRAF mutations by examining the tumour’s DNA using next-generation sequencing or PCR-based methods that detect specific genetic alterations.
Your tumour will be described as BRAF-positive if a mutation is detected or BRAF-negative if no mutation is found.
BRCA1 and BRCA2
BRCA1 and BRCA2 are genes involved in repairing damaged DNA. Mutations in these genes can be inherited or arise only in the tumour. These biomarkers are important because patients with BRCA1 or BRCA2 mutations may benefit from PARP inhibitors, a type of targeted therapy that exploits the tumour’s reduced ability to repair DNA. Identifying these mutations also has implications for family members who may carry the same inherited mutation.
Testing is performed using next-generation sequencing to analyze both inherited genetic material (germline testing) and tumour-specific DNA changes (somatic testing).
Results will state whether the tumour is BRCA1-positive or BRCA2-positive if a mutation is found, or BRCA1-negative or BRCA2-negative if no mutations are detected. Reports may also specify whether the mutation is inherited.
EGFR
EGFR is a protein on the surface of cells that promotes growth and division. Although EGFR is an important biomarker in some cancers, EGFR mutations are uncommon in pancreatic cancer. However, identifying an EGFR alteration may help guide therapy in rare cases.
EGFR testing is performed using next-generation sequencing to look for mutations in the tumour’s DNA.
The tumour will be described as EGFR-positive if a mutation is present or EGFR-negative if no mutation is found.
ERBB2 (HER2)
ERBB2, also known as HER2, is a gene that promotes cell growth. In a small number of pancreatic cancers, HER2 may be overactive. HER2 is important because tumours with HER2 overexpression or amplification may respond to HER2-targeted therapies.
HER2 is tested using immunohistochemistry to evaluate protein expression and, when needed, additional testing such as FISH or next-generation sequencing to identify gene amplification.
HER2 protein levels are scored as 0, 1+, 2+, or 3+. Scores of 0 or 1+ are HER2-negative, 3+ is HER2-positive, and 2+ is equivocal and requires additional testing to confirm gene amplification.
KRAS
KRAS is one of the most commonly mutated genes in pancreatic cancer. Mutations in KRAS drive tumour growth by keeping growth signals turned on continuously. This biomarker is important because KRAS mutations strongly influence tumour behaviour and may determine eligibility for new targeted treatments directed at specific KRAS subtypes.
KRAS testing is performed using next-generation sequencing to evaluate specific regions of the gene where mutations commonly occur.
Your tumour will be described as KRAS-positive if a mutation is found and KRAS-negative if no mutation is detected.
Mismatch repair proteins (MLH1, MSH2, MSH6, PMS2)
Mismatch repair (MMR) proteins help correct small errors in DNA. When any of these proteins are lost, the tumour becomes mismatch-repair deficient (dMMR) and may respond to immunotherapy. MMR deficiency may also indicate Lynch syndrome, an inherited condition that increases the risk of several types of cancer.
Pathologists use immunohistochemistry to determine whether each MMR protein is present in tumour cell nuclei.
The tumour will be described as MMR-proficient (pMMR) if all proteins are present or MMR-deficient (dMMR) if any are absent.
PALB2
PALB2 is a gene that works closely with BRCA1 and BRCA2 in DNA repair. PALB2 mutations can be inherited or occur only in the tumour. This biomarker is important because PALB2-mutated tumours may respond to PARP inhibitors, similar to BRCA-associated cancers.
Testing is performed using next-generation sequencing to identify mutations in the PALB2 gene.
Your tumour will be described as PALB2-positive if a mutation is detected and PALB2-negative if no mutation is found.
RAD51C and RAD51D
RAD51C and RAD51D are genes involved in repairing damaged DNA. Mutations in these genes may indicate that the tumour is sensitive to PARP inhibitors, making them important biomarkers in treatment planning.
These genes are analyzed using next-generation sequencing, which evaluates the DNA for specific mutations.
The tumour will be described as RAD51C-positive or RAD51D-positive if mutations are present and negative if no mutations are detected.
RET fusions
RET is a gene that can fuse with another gene, creating an abnormal fusion protein that promotes tumour growth. Although rare, RET fusions are important because tumours with this change may respond to RET-targeted therapies.
RET fusions are typically detected using next-generation sequencing or FISH to identify gene rearrangements.
The tumour will be described as RET-positive if a gene fusion is detected and RET-negative if no fusion is found.
What is the prognosis for ductal adenocarcinoma of the pancreas?
Ductal adenocarcinoma is an aggressive cancer with a high chance of spreading or coming back after treatment. The prognosis depends on the stage of the tumour at diagnosis, whether it was completely removed with surgery, and whether it has spread to lymph nodes or other organs. The five-year survival rate is low, but surgery combined with chemotherapy may improve outcomes for some patients.
Questions to ask your doctor
-
What was the size and stage of the tumour?
-
Were any lymph nodes involved?
-
Were the margins clear?
-
Was there evidence of perineural or lymphovascular invasion?
-
Are any additional treatments recommended?
-
Was molecular testing performed on my tumour?


