by Jason Wasserman MD PhD FRCPC
February 4, 2026
Hürthle cell carcinoma is a rare type of thyroid cancer that arises from follicular cells, which produce thyroid hormone. This tumour is composed mainly of Hürthle cells (also called oncocytic cells), which are larger than normal thyroid cells and appear pink and granular under the microscope because they contain many mitochondria. These structures produce energy for the cell.
Hürthle cell carcinoma is now more accurately referred to as oncocytic carcinoma in modern pathology. Both terms describe the same disease, but oncocytic carcinoma is preferred because it better reflects the tumour’s biology. You may still see the term Hürthle cell carcinoma used in medical records, imaging reports, or older pathology reports.
This article explains how Hürthle cell carcinoma is diagnosed, classified, and examined under the microscope, and how these findings relate to prognosis and follow-up.
Where does Hürthle cell carcinoma arise?
Most Hürthle cell carcinomas arise within the thyroid gland. Rarely, tumours can develop in ectopic thyroid tissue, meaning thyroid tissue that formed outside its usual location during development. Examples include the tongue (lingual thyroid) or the chest (mediastinum).
What are the symptoms of Hürthle cell carcinoma?
Many patients notice a slowly enlarging, painless thyroid nodule. Tumours with limited invasion are often discovered incidentally during imaging or routine physical examination.
Larger or more invasive tumours may cause:
-
A visible or palpable neck mass.
-
Pressure or tightness in the neck.
-
Difficulty swallowing or breathing.
-
Hoarseness.
Most patients have normal thyroid hormone levels, so symptoms of overactive or underactive thyroid function are uncommon.
How is this diagnosis made?
Diagnosing Hürthle cell carcinoma requires several steps because this tumour cannot be definitively diagnosed by imaging or needle biopsy alone.
The diagnostic process may include:
-
Imaging studies.
-
Surgical removal of the tumour.
-
Careful microscopic examination of the tumour capsule and blood vessels.
Each step provides important information, but the final diagnosis is almost always made after surgery.
Imaging
Ultrasound is usually the first imaging test. Hürthle cell carcinoma cannot be reliably distinguished from benign Hürthle cell adenoma by ultrasound alone. Many tumours appear as solid nodules with a surrounding halo created by the tumour capsule.
Tumours that extend beyond the capsule may show irregular margins or invasion into nearby tissues. Most Hürthle cell carcinomas are hypofunctioning (cold) on radioactive iodine scans, although rare functioning tumours exist. Many are FDG-PET–avid, meaning they take up glucose on PET imaging.
Fine-needle aspiration (FNA)
FNA can identify a Hürthle cell tumour, but it cannot determine whether the tumour is benign or malignant. This is because capsular and vascular invasion cannot be assessed on small biopsy samples.
As a result, FNA reports often use terms such as Hürthle cell neoplasm or suspicious for Hürthle cell neoplasm, and surgical removal is required to establish the diagnosis.
Microscopic features
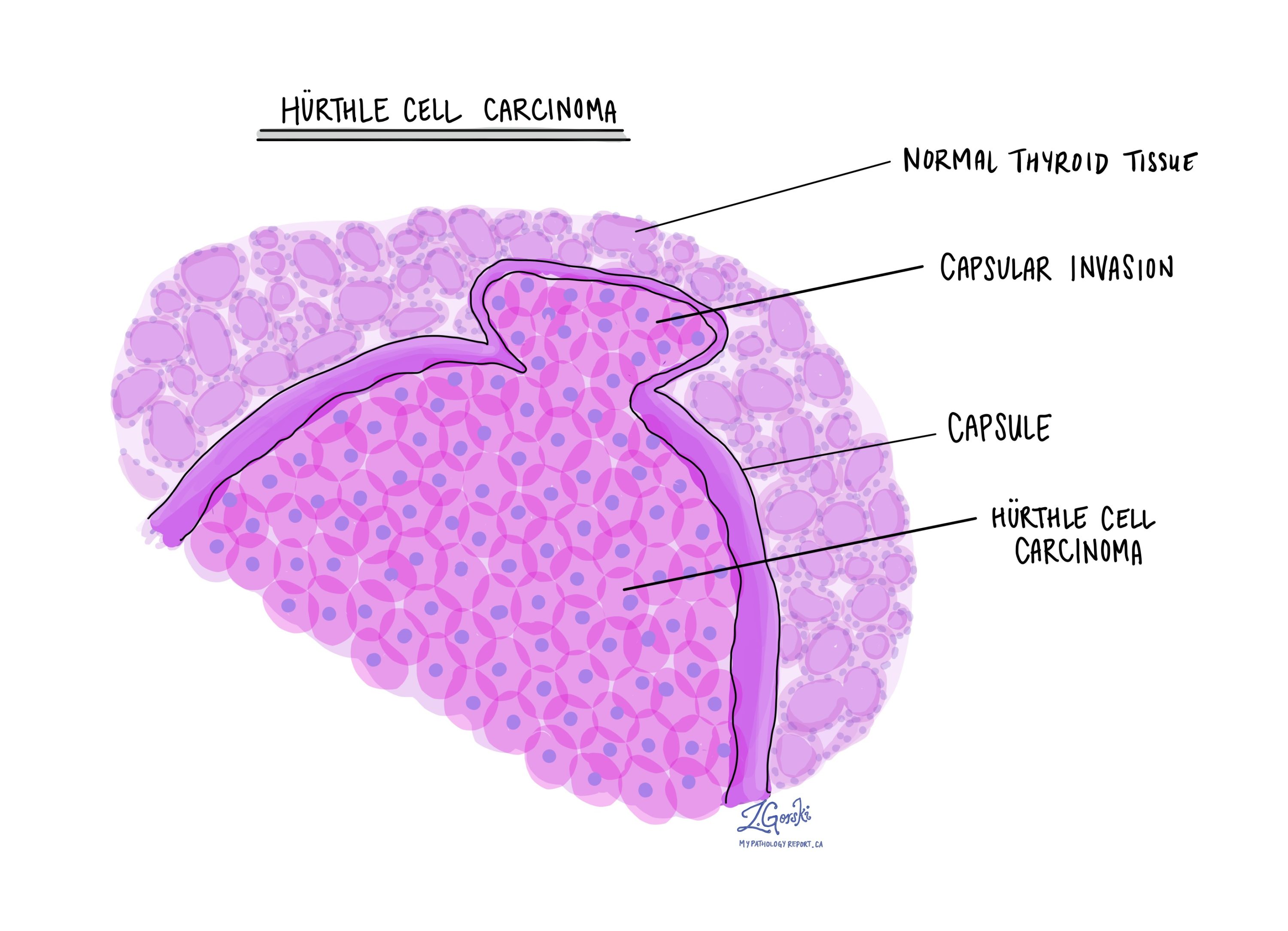
Under the microscope, Hürthle cell carcinoma is typically a well-circumscribed, thickly encapsulated tumour composed of at least 75% Hürthle (oncocytic) cells.
The tumour cells have abundant pink, granular cytoplasm and round nuclei with prominent nucleoli. Growth patterns are commonly solid or trabecular, with fewer follicles than seen in benign tumours. The capsule is usually thicker than that of a Hürthle cell adenoma and may contain calcifications.
The most critical microscopic findings are:
-
Capsular invasion, meaning tumour cells have entirely grown through the capsule.
-
Vascular invasion, meaning tumour cells are found inside blood vessels.
These features confirm that the tumour is malignant and help determine its aggressiveness.
Tumour classification (subtypes)
Hürthle cell carcinoma is subdivided based on how the tumour grows and spreads. This classification is important because it strongly predicts behaviour, recurrence risk, and the likelihood of distant spread.
Minimally invasive Hürthle cell carcinoma
This subtype shows capsular invasion only, without invasion into blood vessels. The tumour remains otherwise well contained.
Minimally invasive tumours usually behave indolently and have an excellent prognosis when completely removed. Additional treatment beyond surgery is often not required.
Encapsulated angioinvasive Hürthle cell carcinoma
These tumours are fully encapsulated but show vascular invasion. Pathologists count the number of involved blood vessels and describe the invasion as:
-
Limited vascular invasion (fewer than 4 vessels).
-
Extensive vascular invasion (4 or more vessels).
Tumours with limited vascular invasion have an intermediate risk, while those with extensive vascular invasion behave more aggressively and require closer follow-up.
Widely invasive Hürthle cell carcinoma
Widely invasive tumours show extensive infiltration into surrounding thyroid tissue or soft tissues, often with multiple areas of vascular invasion.
This subtype has the highest risk of recurrence and distant metastasis, most commonly to the lungs, bones, or liver, and requires more intensive management and long-term surveillance.
Vascular invasion
Vascular invasion means tumour cells are found inside blood vessels, often attached to the vessel wall or mixed with a blood clot. This is one of the most important features in Hürthle cell carcinoma.
Tumours without vascular invasion usually remain localized. In contrast, tumours with vascular invasion—especially when multiple vessels are involved—have a significantly higher risk of spreading to distant organs. Because of this, the presence and extent of vascular invasion strongly influence prognosis and treatment planning.
Extrathyroidal extension
Extrathyroidal extension means tumour cells have grown beyond the thyroid gland into surrounding tissues. A thin layer of connective tissue normally surrounds the thyroid, and most Hürthle cell carcinomas remain confined to the gland.
-
Microscopic extrathyroidal extension can only be seen on microscopic examination and does not change the tumour stage.
-
Macroscopic (gross) extrathyroidal extension is visible during surgery or imaging and involves nearby structures such as neck muscles, the trachea (windpipe), or the esophagus.
Only macroscopic extrathyroidal extension increases the tumour stage because it is associated with a higher risk of recurrence and may affect treatment decisions.
High-grade transformation
In rare cases, Hürthle cell carcinoma can undergo high-grade transformation, meaning it changes into a more aggressive form of thyroid cancer, such as poorly differentiated thyroid carcinoma or high-grade differentiated thyroid carcinoma, oncocytic type.
Features that suggest high-grade transformation include tumour necrosis, increased numbers of dividing cells, abnormal mitotic figures, and loss of typical oncocytic features. Tumours with high-grade transformation are more aggressive, often resistant to radioactive iodine, and have a worse prognosis.
Lymph nodes
Lymph nodes are small immune organs that filter lymphatic fluid. Cancer cells can spread from the thyroid to nearby lymph nodes through lymphatic vessels.
Unlike follicular thyroid carcinoma, Hürthle cell carcinoma can involve lymph nodes, though this is less common than spread via the bloodstream. Any lymph nodes removed during surgery are examined under the microscope and reported as positive or negative for tumour cells.
Lymph node involvement may increase the cancer stage and influence recommendations for additional treatment and surveillance.
Pathologic stage (pTNM)
The pathologic stage for Hürthle cell carcinoma is based on the TNM staging system, which considers the primary tumour (T), lymph nodes (N), and distant metastases (M). In general, higher stages indicate more advanced disease and a worse prognosis.
Tumour stage (pT)
-
T1: Tumour ≤ 2 cm, confined to the thyroid.
-
T2: Tumour > 2 cm but ≤ 4 cm, confined to the thyroid.
-
T3: Tumour > 4 cm or extension into nearby muscles.
-
T4: Tumour extends into structures such as the trachea, larynx, or esophagus.
Nodal stage (pN)
-
N0: No tumour cells in lymph nodes.
-
N1a: Tumour cells in central neck lymph nodes (levels 6–7).
-
N1b: Tumour cells in lateral neck lymph nodes (levels 1–5).
-
NX: No lymph nodes examined.
Prognosis and prediction
The prognosis for Hürthle cell carcinoma depends mainly on the extent of invasion, especially vascular invasion.
Tumours with capsular invasion only have an excellent outcome. Tumours with limited vascular invasion have an intermediate risk, while those with extensive vascular invasion or wide invasion have a significantly worse prognosis.
A small percentage of tumours may later transform into anaplastic thyroid carcinoma, either at recurrence or at initial diagnosis, which carries a very poor prognosis.
Questions to ask your doctor
-
Was my tumour minimally invasive, angioinvasive, or widely invasive?
-
Was vascular invasion present, and how extensive was it?
-
Do I need additional treatment such as radioactive iodine?
-
What is my risk of recurrence or spread?
-
How often will I need follow-up imaging or blood tests?


