by Cathryn Lapedis MD, MPH
March 30, 2023
What is membranous glomerulonephritis?
Membranous glomerulonephritis (also called membranous nephropathy or membranous glomerulopathy) is an autoimmune disease that damages the small filtering units of the kidney called the glomeruli. This damage leads to high levels of protein in the urine.
What do the kidneys do?
The kidneys are paired bean-shaped organs located just below the ribs in the back of the abdomen and close to the spine. The kidneys’ most important function is to filter your blood. Removing waste products from the blood helps control your body’s electrolytes (sodium, potassium, and calcium) and water. These waste products and extra water are made into urine, which flows from the kidneys into the bladder.
The job of filtering the blood takes place in a part of the kidney called the nephron, and to make sure the job gets done, each kidney has millions of nephrons. At the heart of each nephron is a round structure called the glomerulus (multiple are called glomeruli). Blood enters the glomerulus through a small blood vessel called an arteriole which then splits up into many even smaller vessels called capillaries. Inside the glomeruli are specialized mesangial cells, which support the capillaries. Surrounding the capillaries and mesangial cells in the glomerulus is a crescent moon-shaped structure called Bowman’s capsule. The cells that cover the surface of Bowman’s capsule are called podocytes and they are very important because they help decide what needs to stay in the blood and what needs to be removed.
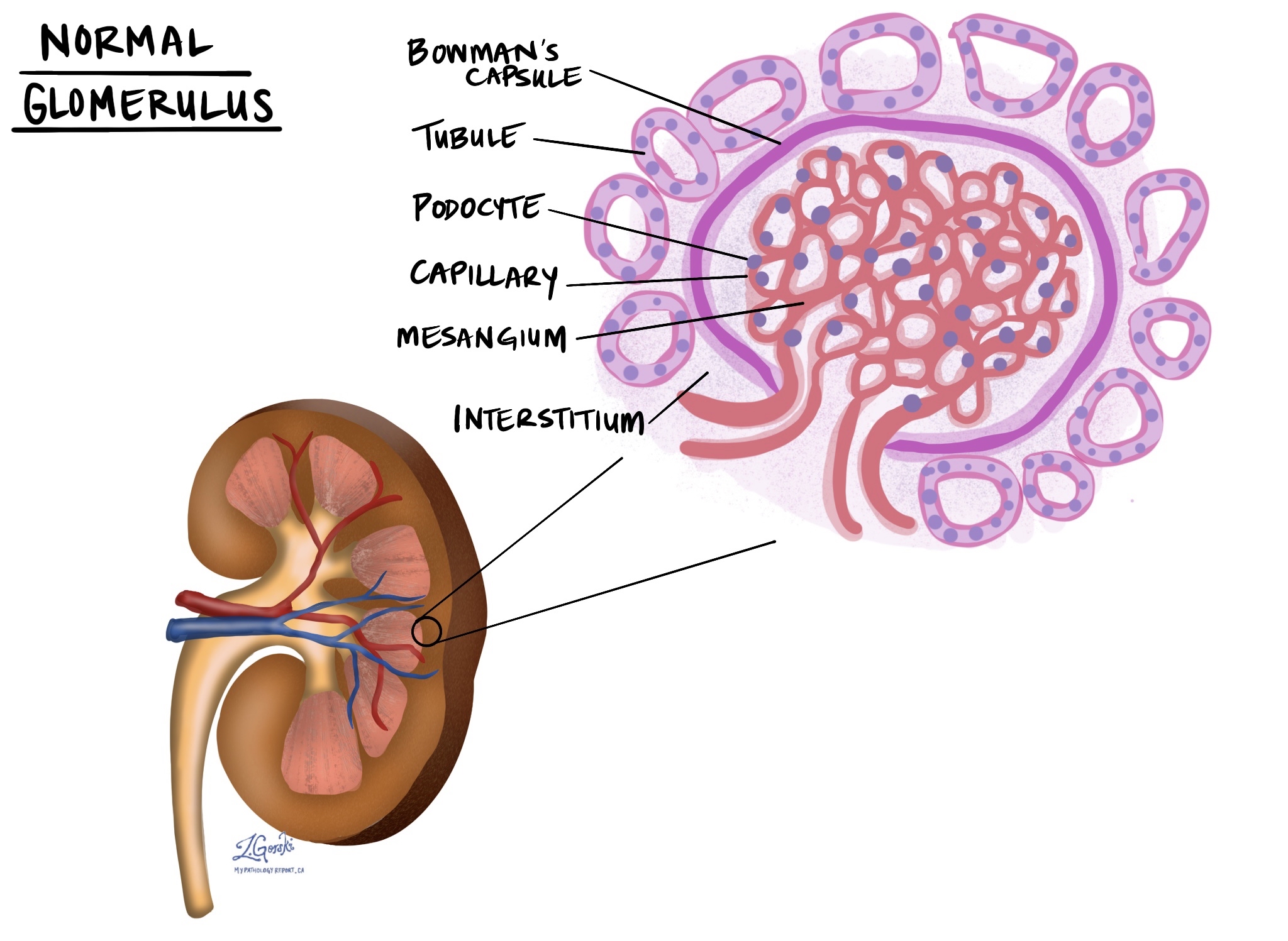
Waste products and extra water that are allowed to cross the podocytes enter a space within Bowman’s capsule. Once inside Bowman’s capsule, the waste products and water are called filtrate. From Bowman’s capsule, the filtrate flows into a long, thin tube called the renal tubule, which returns some of the electrolytes and water into the blood. The rest of the filtrate is turned into urine. The millions of nephrons in the kidney are held together by a special type of connective tissue called the interstitium.
What causes membranous glomerulonephritis?
Membranous glomerulonephritis is caused by autoantibodies that stick to antigens (proteins) on podocytes in the glomerulus. Antibodies (immunoglobulins) are small molecules made by immune cells called plasma cells. Antibodies are designed to stick to foreign antigens such as those found on viruses and bacteria. Autoantibodies are abnormal because they stick to tissues normally found in our body (‘auto’ means self).
Once the autoantibody sticks to the antigen on the podocyte it forms an antibody-antigen immune complex. Over time, large numbers of these immune complexes lump together forming a deposit. In membranous glomerulonephritis, the deposits accumulate around the outside of the capillary loop (in a spot called the subepithelial space) in the glomerulus. These deposits damage the podocytes and the glomerular basement membrane. Because the podocytes are responsible for keeping large molecules such as proteins in the blood while making urine, the damage caused by the immune complex deposits allows large amounts of protein to leak from the blood into the urine.
What are the symptoms of membranous glomerulonephritis?
People with membranous glomerulonephritis may notice that their urine looks bubbly or frothy. This is caused by high levels of protein in the urine. Conversely, low levels of protein in the blood can lead to edema (swelling) around the eyes, ankles, and feet.
What will blood and urine tests show in people with membranous glomerulonephritis?
When blood tests are performed, people with membranous glomerulonephritis will often have low levels of albumin (a protein normally found in the blood) and high levels of lipids (fats). Urine tests will show high levels of protein.
What does nephrotic syndrome mean and how is it related to membranous glomerulonephritis?
Nephrotic syndrome is used to describe the combination of high levels of protein in the urine, low levels of albumin in the blood, and edema (swelling). Nephrotic syndrome is common in people with membranous glomerulonephritis.
What are immune deposits and why are they important?
Immune deposits are antibody-antigen complexes (collections of antibodies and antigens stuck together) that can be seen microscopically by immunofluorescence microscopy or electron microscopy. Immune deposits help pathologists make the diagnosis of membranous glomerulonephritis.
What is the glomerular basement membrane and what does it do?
The glomerular basement membrane is the wall of the capillary in the glomerulus. It separates the blood from the urine. In the kidney, the basement membrane has special properties that allow certain molecules to pass through it while blocking other molecules. It also acts as structural support for two types of cells, endothelial cells on the blood side of the glomerular basement membrane, and podocytes on the urine side of the glomerular basement membrane.
What does it mean if the glomerular basement membrane is thickened?
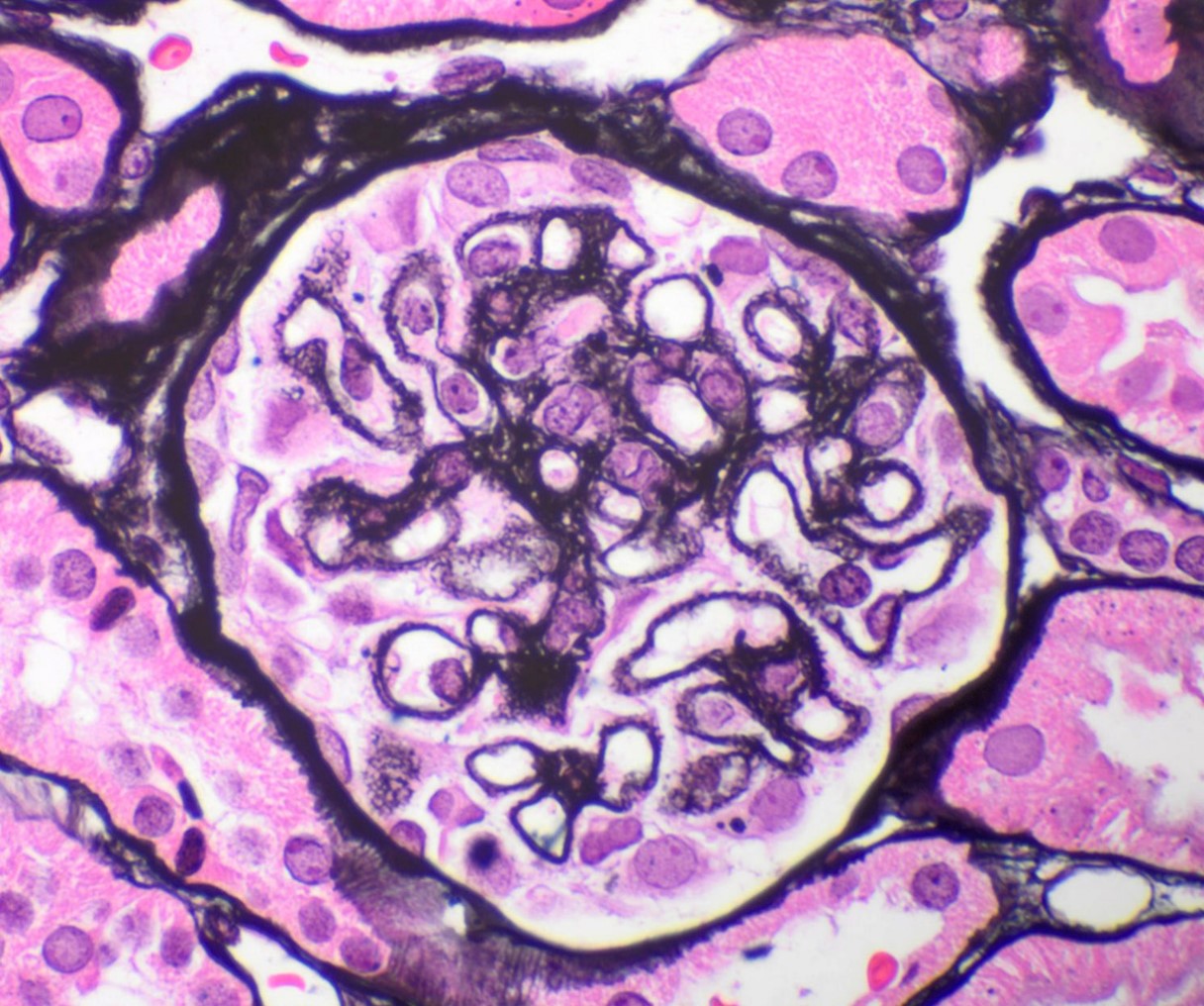
A thickened glomerular basement membrane results from repeated attempts by the body to repair a damaged basement membrane. In membranous glomerulonephritis the thickened glomerular basement membrane takes the form of spikes and craters which gives it a fuzzy appearance when examined using a special silver stain under the microscope.
What does it mean if there are spikes in the basement membrane?
In membranous glomerulonephritis, spikes are formed when the basement membrane grows up and around the immune complex deposits. Spikes can be seen on the silver stain via light microscopy and electron microscopy.

What is the Ehrenreich and Churg staging system and why is the stage important for patients with membranous glomerulonephritis?
Pathologists use the Ehrenreich and Churg staging system to describe the changes normally seen in membranous glomerulonephritis. The system has four stages with stage I representing the earliest changes and stage IV representing changes seen late in the disease.
- Stage I: Small immune complex deposits sit right along the subepithelial space of the basement membrane in between the podocyte and the basement membrane. There is no basement membrane reaction or thickening.
- Stage II: Immune complex deposits are present in the subepithelial space, and the basement membrane begins to grow up around them forming “spike-like” projections.
- Stage III: Immune complex deposits are present in the subepithelial space. The basement membrane has expanded to surround the deposits.
- Stage IV: The immune complex deposits are fully surrounded by basement membrane. Later the deposits may be absorbed and disappear.
What is light microscopy and why is it performed?
Light microscopy is a technique where tissue is examined under a microscope with visible light. Light microscopy allows pathologists to see very small structures in the kidneys such as tubules, glomeruli, and blood vessels.
What is immunofluorescence microscopy and why is it performed?
Immunofluorescence microscopy is a type of microscopic examination that uses fluorescent light (instead of the visible light used on a regular microscope). The fluorescent light makes it easier for pathologists to see antibodies and other molecules made by the immune system inside tissue and to recognize patterns of disease. For example, in a condition called PLA2R associated- membranous glomerulonephritis we expect to see reactivity for the immune molecules IgG, C3, and PLA2R.

What is electron microscopy and why is it performed?
Electron microscopy is a type of microscopic examination that uses a beam of electrons to look inside tissue. By using electrons instead of light, electron microscopes can see parts of cells and tissues that are far too small for regular microscopes. In the kidney, this allows pathologists to see the parts of a podocyte, immune complexes deposited in the tissue, and even viruses inside a cell.

What does glomerulosclerosis mean and why is it important?
Glomerulosclerosis means scarring of the glomerulus (functional unit of the kidney). It is normal to see a small amount of glomerulosclerosis as a person ages (think of it as the wrinkles of aging for your kidney). However, normal age-related glomerulosclerosis should not exceed 5 to 10% of all glomeruli. Higher levels of glomerulosclerosis is a sign of long-standing membranous glomerulonephritis.
In general, when more than 50% of glomeruli are scarred, the kidney is less likely to be able to recover fully from the disease. When this happens your nephrologist may start talking to you about filtering the blood with dialysis or kidney transplantation.
What does arterial sclerosis mean and why is it important?
Arterial sclerosis is a type of damage that affects large blood vessels called arteries. Arteries are important because they bring blood to the kidneys. When arterial sclerosis occurs, the kidneys are not able to get enough blood which damages the glomeruli and tubules. Over time, this can lead to poor kidney function.
What does tubular atrophy mean and why is it important?
Tubular atrophy is a term pathologists use to describe scarred or damaged renal tubules. Because the renal tubules are important for moving filtrate out of the glomerulus or glomeruli and making urine, tubular atrophy prevents the glomerulus or glomeruli from functioning normally. Pathologists describe the amount of tubular atrophy as a percentage, for example, 5-10% (very minimal tubular scarring), 10-25% (mild tubular scarring), 26-50% (moderate tubular scarring), and 50% or more (severe tubular scarring). A kidney that shows a high percentage of tubular atrophy is less likely to heal over time.
What does interstitial fibrosis mean and why is it important?
Interstitial fibrosis is a type of scar that forms in a part of the kidney called the interstitium. Because the interstitium helps hold the millions of nephrons in the kidney together, interstitial fibrosis makes it difficult for the kidney to function normally. Pathologists describe the amount of interstitial fibrosis as a percentage, for example, 5-10% (very minimal interstitial scarring), 10-25% (mild interstitial scarring), 26-50% (moderate interstitial scarring), and 50% or more (severe interstitial scarring). A kidney that shows a high percentage of interstitial fibrosis is less likely to heal over time.
What does it mean if a change is described as “global”?
When examining a kidney biopsy, pathologists use the term global to mean that the whole glomerulus is involved. For example, global glomerulosclerosis means the whole glomerulus is scarred and not working.
What does it mean if a change is described as “segmental”?
When examining a kidney biopsy, pathologists use the term segmental to mean that just part of the glomerulus is involved. For example, segmental glomerulosclerosis means part of the glomerulus is scarred and not working, but other parts of the glomerulus may still be working normally.
What does it mean if a change is described as “focal”?
When examining a kidney biopsy, pathologists use the term focal to mean that the changes were seen in some but not all of the biopsy tissue. For example, focal nodular sclerosis means that some but not all the glomeruli examined show features of nodular sclerosis. Often the term focal will come with a percentage or quantification, which tells you how much of the tissue is involved.
What does it mean if a change is described as “diffuse”?
When examining a kidney biopsy, pathologists use the term diffuse to mean that the changes were seen throughout the entire biopsy tissue. For example, diffuse nodular sclerosis means that all the glomeruli in the biopsy show nodular sclerosis.


