by Jason Wasserman MD PhD FRCPC and Trevor Flood MD FRCPC
November 12, 2025
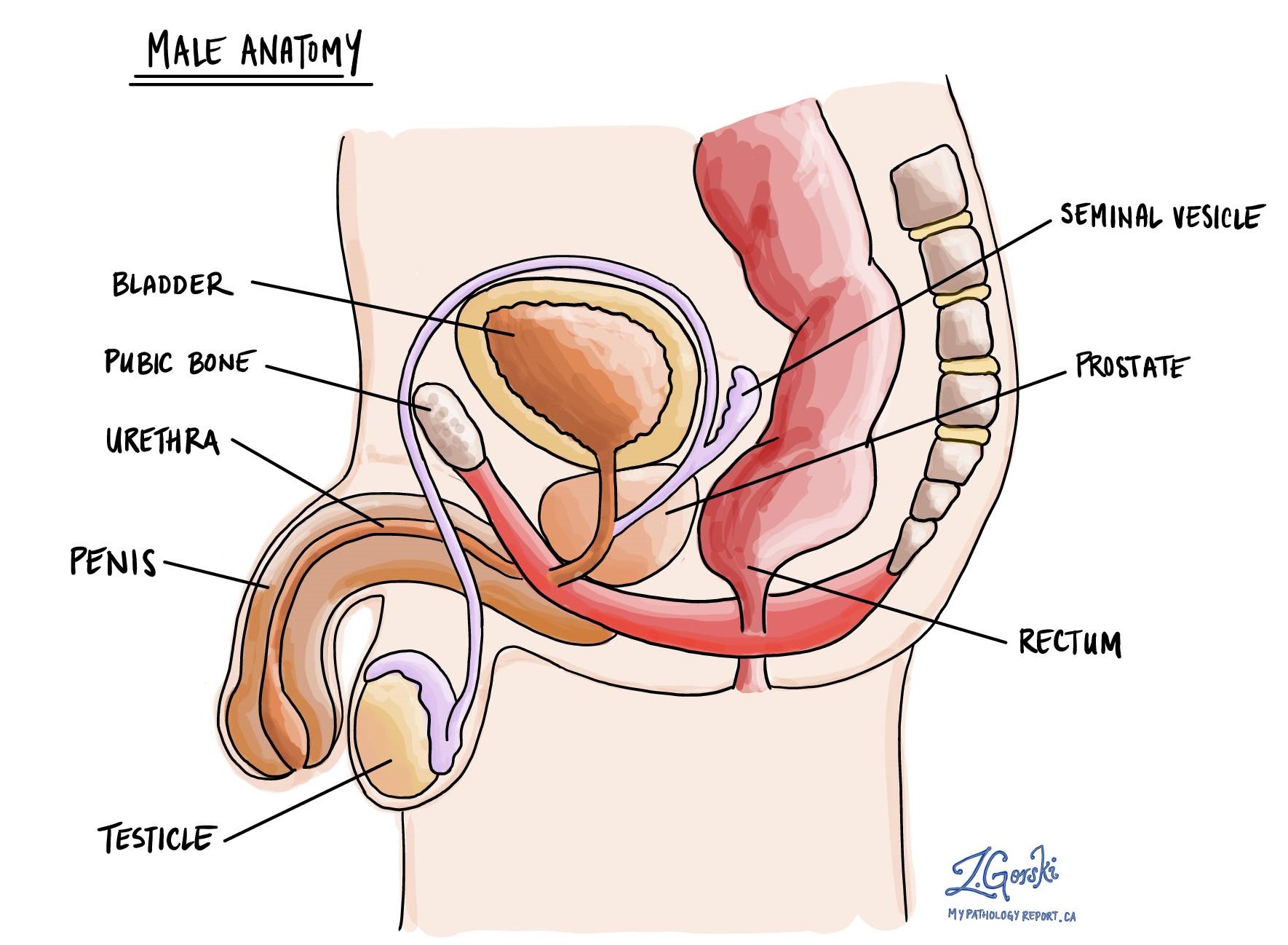
Ductal adenocarcinoma is a rare type of prostate cancer that starts in the glandular cells that line the ducts of the prostate. The prostate gland sits just below the bladder and in front of the rectum. Its normal glands produce and secrete seminal fluid, which mixes with sperm to form semen.
Ductal adenocarcinoma tends to grow in complex patterns and is generally more aggressive than the more common type of prostate cancer, known as acinar adenocarcinoma.
What are the symptoms of ductal adenocarcinoma?
The symptoms of ductal adenocarcinoma are similar to those of other types of prostate cancer. They include:
-
Difficulty starting or maintaining urination.
-
Weak urine flow or dribbling.
-
Blood in the urine or semen.
-
Pain or discomfort in the lower back, pelvis, or hips.
Some people may not have symptoms at all, particularly in the early stages of the disease.
What causes ductal adenocarcinoma?
The exact cause of ductal adenocarcinoma is not fully understood. Like other types of prostate cancer, it is thought to result from a combination of genetic factors, hormonal influences, and environmental exposures. The risk of developing ductal adenocarcinoma increases with age and family history of prostate cancer.
The same risk factors associated with acinar adenocarcinoma also apply to this type of cancer. Ductal adenocarcinoma may arise in areas of prostatic intraepithelial neoplasia, a precancerous condition in which prostate gland cells start to show abnormal growth but have not yet become invasive cancer.
What is the difference between ductal adenocarcinoma and acinar adenocarcinoma?
Both ductal and acinar adenocarcinomas arise from glandular cells in the prostate, but they differ in their microscopic appearance and behaviour.
Acinar adenocarcinoma is the most common type and typically forms small, round glands that resemble normal prostate tissue.
Ductal adenocarcinoma forms tall, column-shaped cells arranged in complex patterns, often in larger ducts near the urethra. It is usually diagnosed at a more advanced stage and has a higher risk of spreading outside the prostate.
Mixed ductal and acinar adenocarcinoma
Ductal adenocarcinoma often occurs in combination with the more common acinar adenocarcinoma. When this happens, the cancer may be described as prostatic adenocarcinoma with ductal features.
If the ductal pattern makes up more than half of the tumour after the prostate is removed, the diagnosis is changed to ductal adenocarcinoma. Even when it is present in smaller amounts, the ductal component can influence the tumour’s behaviour, making it more aggressive and affecting treatment decisions.
How is this diagnosis made?
The diagnosis of ductal adenocarcinoma is made after a pathologist examines a biopsy. Most prostate cancers are discovered when a digital rectal examination detects an abnormality or when a blood test shows an elevated level of prostate-specific antigen (PSA).
Biopsy
A core needle biopsy of the prostate involves taking several small tissue samples from different parts of the gland, typically 10 to 15 samples. These samples are placed in fixative and sent to the pathology laboratory for microscopic examination.
Microscopic examination
Under the microscope, the pathologist looks for abnormal glandular structures made up of tall, column-shaped cells with large, dark nuclei and prominent nucleoli. These cells often form papillary (finger-like) or cribriform (sieve-like) patterns, which are characteristic of ductal adenocarcinoma. Areas of comedonecrosis, meaning clusters of dead cells surrounded by living tumour cells, may also be present.
If only a biopsy sample is available, the pathologist will describe these features and assign a Gleason grade to indicate the aggressiveness of the tumour. Other features such as tumour size, extraprostatic extension, and margin status can only be assessed after the entire prostate is surgically removed.
After surgery, the pathologist examines the prostate, seminal vesicles, and any lymph nodes removed. The final report includes the percentage of ductal and acinar components, the Gleason score, the presence of extraprostatic extension or invasion into nearby organs, and whether all cancer has been removed.
What is the Gleason grade and score?
The Gleason grading system is used to describe the microscopic growth pattern of prostate cancer. Each tumour is assigned two grades, each ranging from 1 to 5, based on the most common and the second most common growth patterns observed. These numbers are then combined to produce a Gleason score, ranging from 6 to 10. Higher scores indicate more aggressive cancers.
Ductal adenocarcinoma is typically assigned a Gleason grade of 4 because it forms complex glandular structures. When comedonecrosis is present, the tumour is given a Gleason grade of 5, reflecting even more aggressive behaviour.
Because ductal adenocarcinoma typically has a higher Gleason score, it tends to grow and spread more rapidly than lower-grade acinar adenocarcinomas.
Tumour quantification
Tumour quantification describes how much of the prostate is replaced by cancer cells. The pathology report includes:
-
The number of biopsy cores that contain cancer.
-
The percentage of each core that is involved by the tumour.
After surgery, the pathologist may estimate the overall percentage of the prostate replaced by cancer. Tumour quantification provides an estimate of tumour size and helps guide treatment decisions.
Extraprostatic extension
Extraprostatic extension refers to the growth of cancer cells beyond the prostate and into the surrounding fatty tissue or connective tissue. This finding indicates that the cancer has spread outside the gland and is associated with a higher risk of recurrence. Extraprostatic extension is an important factor in determining the pathologic stage.
Seminal vesicle invasion
The seminal vesicles are two small glands located behind the bladder and above the prostate. They produce part of the fluid that nourishes and transports sperm. Seminal vesicle invasion means cancer cells have spread directly from the prostate into these glands. This finding indicates more advanced disease and is associated with a higher tumour stage and worse prognosis.
Bladder neck invasion
The bladder neck is the lower part of the bladder that connects to the prostate. Bladder neck invasion means that cancer cells have spread from the prostate into this area. It is considered an advanced feature and is included in determining the tumour stage.
Perineural invasion
Perineural invasion (PNI) means cancer cells are growing along or around a nerve. Nerves are small structures that carry signals for sensation and movement. When tumour cells travel along nerves, they can spread further within the pelvis and may increase the risk of recurrence after surgery. Perineural invasion is a common finding in prostate cancer and is included in the pathology report because it helps assess prognosis.
Lymphovascular invasion
Lymphovascular invasion (LVI) means cancer cells are seen inside blood vessels or lymphatic channels near the tumour. Blood vessels carry blood, while lymphatic channels carry a clear fluid called lymph to lymph nodes. Finding cancer in these vessels means that the tumour has the potential to spread to lymph nodes or distant organs.
Lymphovascular invasion is associated with a higher risk of recurrence and is used to help guide postoperative treatment planning.
Margins
Margins refer to the edges of tissue removed during surgery. After prostate removal, the pathologist examines all surfaces to determine whether cancer cells are present at or near the edge.
A negative margin means no cancer cells are seen at the edge, suggesting that the tumour was removed entirely. A positive margin means cancer cells are found at the edge, suggesting that some cancer may remain in the body. The distance between the tumour and the margin may also be recorded. Margin status is an important indicator of whether additional treatment may be needed.
Lymph nodes
Lymph nodes are small immune organs that filter fluid and trap abnormal cells, including cancer cells. The prostate drains into lymph nodes in the pelvis, which are often removed during surgery to check for cancer spread.
The pathologist examines each lymph node under the microscope to see if it contains cancer cells. The report includes the total number of lymph nodes examined, the number that contain cancer, and whether the cancer has grown beyond the capsule of the lymph node into nearby tissue (known as extranodal extension).
The presence of cancer in lymph nodes increases the risk that the disease may spread elsewhere in the body and affects the nodal stage (pN).
Pathologic stage (pTNM)
The pathologic stage for ductal adenocarcinoma of the prostate is based on the TNM staging system, an internationally recognized system created by the American Joint Committee on Cancer. This system uses information about the primary tumour (T), lymph nodes (N), and distant metastatic disease (M) to determine the complete pathologic stage (pTNM). Generally, a higher number indicates a more advanced disease and a poorer prognosis.
Tumour stage (pT)
Your pathologist will assign a tumour stage between T2 and T4 based on how far the cancer has spread outside the prostate:
-
T2 – The tumour is limited to the prostate.
-
T3 – Cancer has spread outside the prostate into surrounding fat, seminal vesicles, or the bladder neck.
-
T4 – Cancer has spread into nearby organs or tissues such as the rectum or pelvic wall.
Nodal stage (pN)
The nodal stage is based on whether cancer cells are found in lymph nodes:
-
NX – No lymph nodes were submitted for examination.
-
N0 – No cancer cells are found in any lymph nodes examined.
-
N1 – Cancer cells are found in at least one lymph node.
Finding cancer in lymph nodes or in tissues outside the prostate increases the stage and influences treatment planning.
What happens after the diagnosis?
After diagnosis, your healthcare team will review your pathology report, PSA levels, imaging results, and overall health to determine the best treatment plan. The team may include a urologist, an oncologist, a radiation oncologist, and a pathologist.
For most patients, treatment options include surgery (radical prostatectomy) to remove the prostate and surrounding tissue, radiation therapy, or androgen deprivation therapy (hormone therapy) to lower testosterone levels and slow tumour growth. In advanced cases, chemotherapy or targeted therapy may be used.
The presence of ductal adenocarcinoma is important because it can indicate a more aggressive form of prostate cancer. Even when only a small ductal component is found, your doctor will consider this feature when recommending treatment.
After treatment, you will have regular follow-up appointments to monitor your PSA levels and check for recurrence. Imaging studies may be performed if PSA levels rise or new symptoms develop. Maintaining a healthy lifestyle and attending scheduled follow-ups are important parts of long-term care.
Questions for your doctor
-
What percentage of my tumour shows ductal features?
-
What is my Gleason score, and how does it affect my treatment?
-
Was there extraprostatic extension, seminal vesicle invasion, or bladder neck invasion?
-
Did my tumour show perineural or lymphovascular invasion?
-
Were the surgical margins negative, and was the tumour completely removed?
-
Were lymph nodes removed, and did any contain cancer?
-
What is my pathologic stage (pT and pN categories)?
-
What treatment options do you recommend, and what are the possible side effects?
-
How will my PSA levels be monitored after treatment?
-
How often will I need follow-up visits or imaging?


