by Jason Wasserman MD PhD FRCPC
April 6, 2026
Invasive lobular carcinoma (ILC) is the second most common type of breast cancer after invasive ductal carcinoma. It starts in the lobules — the small glands in the breast that produce milk — and grows into the surrounding breast tissue. The term “invasive” means the cancer cells have spread beyond the lobules into nearby tissue. Under the microscope, invasive lobular carcinoma has a distinctive appearance: the tumor cells are loosely connected and tend to travel as single cells in single-file lines through the breast tissue, rather than forming the glandular clusters typical of invasive ductal carcinoma.
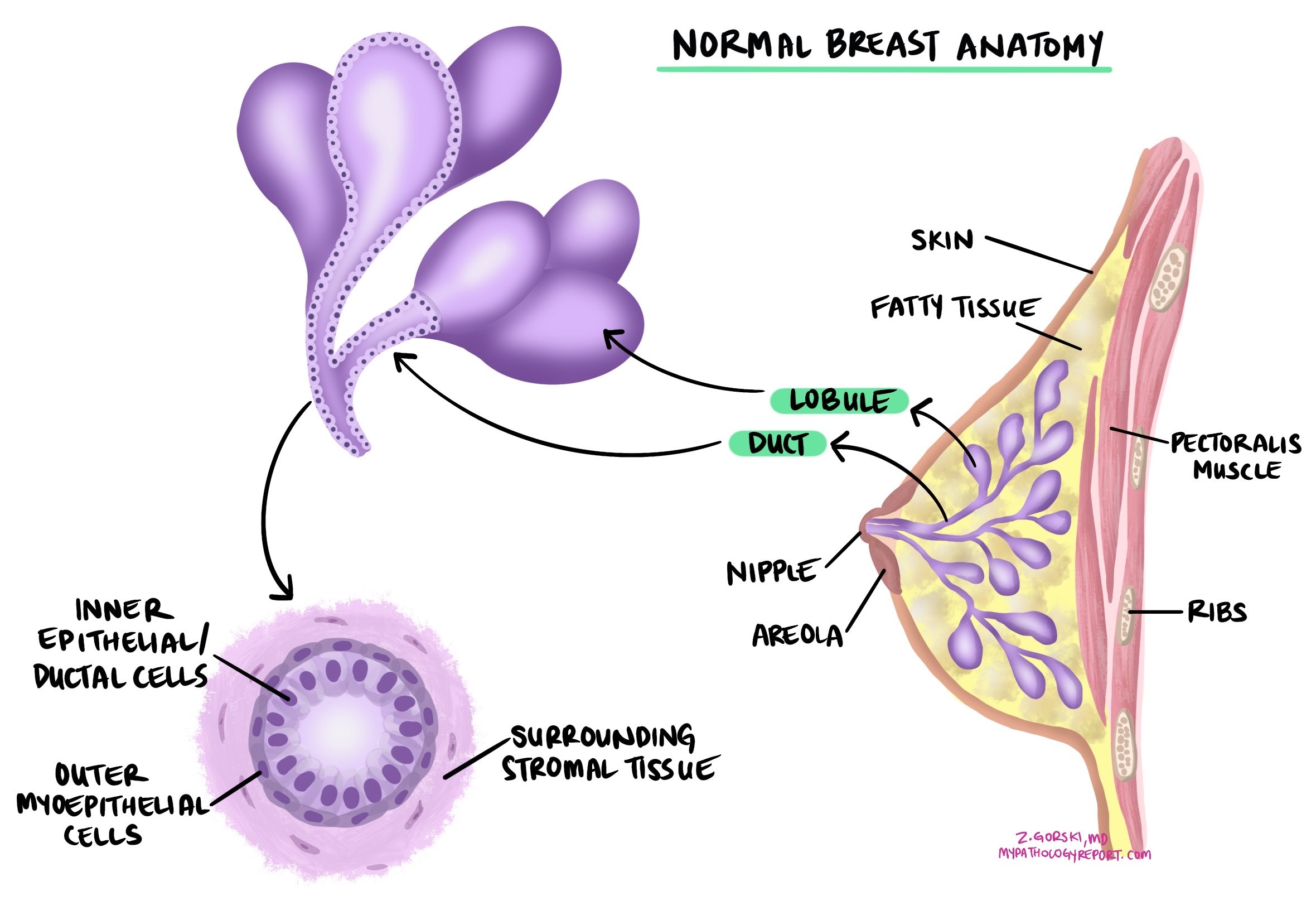
In some patients, invasive lobular carcinoma develops from a precancerous condition called lobular carcinoma in situ (LCIS), in which abnormal cells are confined to the lobules. People with a prior diagnosis of LCIS have a higher risk of developing invasive lobular carcinoma in either breast.
This article will help you understand the findings in your pathology report. If you had a breast biopsy or surgery, you may also find our guide to understanding your breast biopsy report helpful.
What causes invasive lobular carcinoma?
Invasive lobular carcinoma develops due to a combination of genetic, hormonal, and lifestyle factors. The most important genetic driver is the loss of a protein called E-cadherin, which normally helps breast cells stick together. When E-cadherin is lost — most commonly due to mutations or silencing of the CDH1 gene — the cells lose their cohesion and spread individually through the breast tissue, creating the characteristic single-file growth pattern that defines invasive lobular carcinoma.
Inherited CDH1 gene mutations are associated with hereditary diffuse gastric cancer syndrome and also significantly increase the lifetime risk of invasive lobular carcinoma, up to approximately 42% in affected individuals. Genetic counseling is recommended for anyone with a personal or family history suggesting this syndrome. Other commonly altered genes in invasive lobular carcinoma include PIK3CA, PTEN, and RUNX1. In rare cases, HER2 or AKT1 mutations are present and may influence treatment decisions.
Hormonal factors — such as prolonged estrogen exposure through late menopause, hormone replacement therapy, or having no children — also increase risk. Most invasive lobular carcinomas are hormone receptor-positive, reflecting this hormonal dependency.
What are the symptoms?
Because invasive lobular carcinoma tends to grow in a diffuse, infiltrating pattern rather than forming a single distinct mass, it can be harder to detect than invasive ductal carcinoma. Early stages may cause no noticeable symptoms. As the tumor grows, a person may feel a thickening or fullness in the breast rather than a discrete lump, notice changes in the size or shape of the breast, see dimpling of the skin, or experience nipple inversion. Invasive lobular carcinoma is sometimes found on a mammogram or breast MRI before symptoms appear.
How is the diagnosis made?
The diagnosis is usually made after a small sample of the tumor is removed in a biopsy and examined under a microscope by a pathologist. Additional surgery to remove the entire tumor is typically recommended after the biopsy confirms cancer.
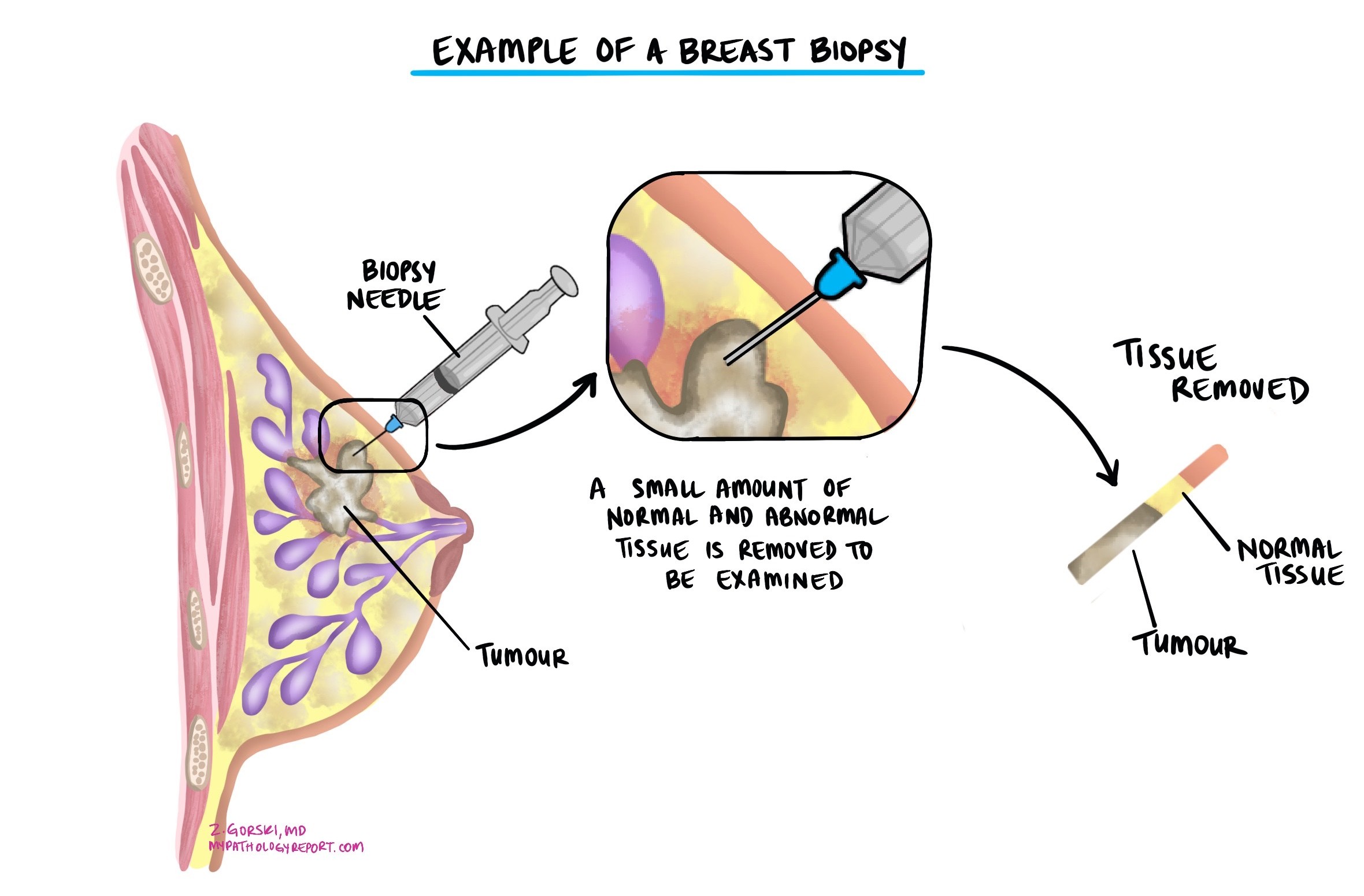
Under the microscope, the pathologist identifies the distinctive single-file growth pattern and dyscohesive (non-clumping) cells. Because invasive lobular carcinoma can look different from invasive ductal carcinoma and may be confused with other tumors, immunohistochemistry — special staining tests that detect specific proteins — is often performed to confirm the diagnosis (see Immunohistochemistry section below).
Histologic types
Pathologists classify invasive lobular carcinoma into subtypes based on the appearance of the cells under the microscope. The subtype may appear in your pathology report.
- Classic type — The most common subtype. The cancer cells are small and uniform, traveling through the breast tissue as individual cells in single-file lines. This subtype is most often low-grade and hormone receptor-positive, and is associated with a more favorable prognosis.
- Pleomorphic type — The cancer cells are larger and more abnormal-looking than the classic type. The nuclei are larger, more irregular, and appear darker (hyperchromatic). This subtype is more likely to be higher grade and may be more aggressive, with a higher risk of spread to lymph nodes and other organs.
- Other variants — Solid, alveolar, mixed, and tubulo-lobular patterns may also be described. These are less common and are generally managed similarly to the classic type.
Nottingham histologic grade
The Nottingham histologic grade assesses how aggressive the tumor is likely to be by scoring three microscopic features, each on a scale of 1 to 3:
- Tubule formation — The proportion of the tumor that forms round, gland-like structures. Invasive lobular carcinoma typically forms very few tubules, so this category usually receives a score of 3.
- Nuclear pleomorphism — How variable and abnormal the nuclei look compared to normal cells. A score of 1 means the nuclei are relatively uniform; a score of 3 means they are markedly enlarged and irregular.
- Mitotic count — How many cells are actively dividing (mitotic figures) in a defined area of the tumor.
The three scores are added together (total range 3–9) to determine the overall grade:
- Grade 1 (low grade) — Total score 3–5. Slower growing, less likely to spread. Most classic-type ILCs are grade 1.
- Grade 2 (intermediate grade) — Total score 6–7: moderate growth rate and risk.
- Grade 3 (high grade) — Total score 8–9. More aggressive, faster growing. More common in the pleomorphic subtype.
Note that because invasive lobular carcinoma rarely forms tubules, most tumors receive a tubule score of 3 regardless of overall aggressiveness — meaning the grade is largely determined by nuclear pleomorphism and mitotic count.
Tumor size
Tumor size is used to determine the pathologic tumor stage (pT) and is an important predictor of outcome — larger tumors are more likely to metastasize to lymph nodes and other organs. The final tumor size can only be accurately measured after the entire tumor has been removed surgically. It will not be included in a biopsy report.
Invasive lobular carcinoma can be challenging to measure accurately because it does not always form a discrete mass — its diffuse growth pattern can make the true extent of the tumor appear larger than it appears on imaging. Breast MRI is particularly useful for assessing the size of invasive lobular carcinoma before surgery.
Tumor extension
Invasive lobular carcinoma begins inside the breast, but in some cases the tumor spreads into the overlying skin or the muscles of the chest wall. This is called tumor extension. Its presence is associated with a higher risk of local recurrence and distant spread, and it raises the pathologic tumor stage (pT4).
Immunohistochemistry
Immunohistochemistry uses special stains to detect specific proteins in tumor cells. For invasive lobular carcinoma, these tests serve two purposes: confirming the diagnosis and providing biomarker information that guides treatment. The most important marker in this context is E-cadherin.
- E-cadherin — This protein, which helps cells stick together, is absent in virtually all invasive lobular carcinomas. This loss is a defining feature of the diagnosis and helps the pathologist distinguish invasive lobular carcinoma from invasive ductal carcinoma (which typically retains E-cadherin). Your report will state that E-cadherin expression is “lost” or “absent” in the tumor cells.
- Estrogen receptor (ER) and progesterone receptor (PR) — Tested as part of standard biomarker assessment (see Biomarker testing section below).
- HER2 — Tested as part of standard biomarker assessment (see below).
Lymphovascular invasion
Lymphovascular invasion means cancer cells have entered small blood vessels or lymphatic channels near the tumor. Once inside these channels, cancer cells can travel to lymph nodes or reach distant organs through the bloodstream. Your report will state whether lymphovascular invasion is “present” or “absent.” Its presence increases the risk of spread and recurrence, and may lead your doctor to recommend additional treatment such as chemotherapy or radiation therapy.
Surgical margins
A margin is the edge of tissue removed during surgery. The pathologist examines the margin surfaces to determine whether the entire tumor was removed.
- Negative margin — No tumor cells at the cut edge. Suggests the visible tumor was completely removed.
- Positive margin — Tumor cells present at the cut edge, raising concern that some cancer remains. Additional surgery or radiation is usually recommended.
Even when all margins are negative, the report may include a measurement of how close the nearest tumor cells came to the edge. Margins are assessed only after surgery that removes the entire tumor, not after a biopsy.
Because invasive lobular carcinoma grows diffusely and may not form a well-defined mass, achieving clear margins can sometimes be more challenging than with invasive ductal carcinoma. Your surgeon and pathologist will work together to ensure complete removal is confirmed.
Lymph nodes
Lymph nodes are small immune organs that can trap cancer cells as they spread through the lymphatic system. When breast cancer spreads, it typically moves first to the axillary (underarm) lymph nodes. During surgery, these nodes are removed and examined. Your pathology report will include the total number of lymph nodes examined, the number containing cancer, and the size of any cancer deposits.
There are three levels of lymph node involvement:
- Isolated tumor cells (ITCs) — Clusters no larger than 0.2 mm. Not counted as positive for staging purposes.
- Micrometastasis — Clusters between 0.2 mm and 2 mm. Reported as pN1mi. May slightly increase recurrence risk.
- Macrometastasis — Clusters larger than 2 mm. Associated with a higher risk of distant spread and typically leads to more intensive treatment recommendations.
Your report may also mention extranodal extension, which means cancer has broken through the outer wall of a lymph node into the surrounding tissue — a finding associated with higher recurrence risk. A sentinel lymph node is the first node in the drainage chain from the breast and is typically the first to be tested.
Biomarker and molecular testing
Biomarker testing is an essential part of every invasive lobular carcinoma workup. The results directly determine which treatments are most likely to be effective.
Estrogen receptor (ER) and progesterone receptor (PR)
The vast majority of invasive lobular carcinomas — approximately 95% — are hormone receptor-positive, expressing estrogen receptor (ER) and/or progesterone receptor (PR). This means the cancer cells use the hormones estrogen and progesterone to fuel their growth. Testing is done by immunohistochemistry.
Your report will include the percentage of positive cells (e.g., “90% ER-positive”), staining intensity (weak, moderate, or strong), and possibly an overall score (Allred or H-score). A cancer is considered hormone receptor-positive if ER or PR is present in at least 1% of cells. Hormone receptor-positive cancers respond well to hormone-blocking therapies such as tamoxifen or aromatase inhibitors (anastrozole, letrozole, exemestane), which reduce the risk of recurrence. These treatments are typically given for 5 to 10 years after surgery.
HER2
HER2 amplification is uncommon in invasive lobular carcinoma — the vast majority of ILCs are HER2-negative. When HER2 testing is performed, it follows the same two-step process used for invasive ductal carcinoma:
- Immunohistochemistry (IHC) — measures HER2 protein on tumor cell surfaces, reported as 0, 0+, 1+, 2+, or 3+. Scores of 0 and 1+ are negative; 3+ is positive; 2+ is equivocal and requires further testing.
- In situ hybridization (ISH/FISH) — performed when IHC is 2+ to check for HER2 gene amplification. A positive (amplified) result confirms HER2-positive status.
As with invasive ductal carcinoma, tumors with an IHC score of 1+ or 2+/ISH-negative are classified as HER2-low, which may be eligible for trastuzumab-deruxtecan in the metastatic setting. Tumors with IHC 3+ are HER2-positive and may respond to HER2-targeted therapies such as trastuzumab. In rare cases of ILC with HER2 amplification, treatment is approached similarly to HER2-positive invasive ductal carcinoma.
Genomic testing (gene expression profiling)
Some patients with hormone receptor-positive, HER2-negative invasive lobular carcinoma may be offered genomic tests that analyze gene activity in the tumor to estimate the risk of recurrence and predict whether chemotherapy is likely to add benefit beyond hormone therapy. These tests include the 21-gene recurrence score (Oncotype DX) and the 70-gene signature (MammaPrint). These results may appear in your pathology report or be provided separately.
However, it is important to note that some genomic assays were primarily validated in invasive ductal carcinoma, and their performance in invasive lobular carcinoma remains under study. Your oncologist will discuss whether genomic testing is appropriate for your case and how the result would be interpreted specifically for ILC.
For more information about breast cancer biomarkers, visit our Biomarkers and Molecular Testing section.
Treatment effect and residual cancer burden
If you received chemotherapy, hormone therapy, or targeted therapy before surgery (called neoadjuvant therapy), your pathology report will describe how much tumor remains after treatment.
The Residual Cancer Burden (RCB) index combines the size of the tumor bed, the percentage of remaining cancer cells, and lymph node involvement into a single score:
- RCB-0 (pathologic complete response) — No residual invasive cancer in the breast or lymph nodes. Most favorable result.
- RCB-I (minimal residual disease) — Very little cancer remains.
- RCB-II (moderate residual disease) — A moderate amount of cancer remains.
- RCB-III (extensive residual disease) — A large amount of cancer remains, associated with a higher recurrence risk.
It is worth noting that invasive lobular carcinoma typically shows a lower rate of complete pathologic response to neoadjuvant chemotherapy compared to other breast cancer subtypes. This reflects ILC’s biology — it is usually hormone receptor-positive and grade 1–2, meaning it is less chemotherapy-sensitive. Neoadjuvant hormone therapy is increasingly used as an alternative in some cases.
Pathologic stage (pTNM)
The pathologic stage describes how far the cancer has spread, using the TNM staging system. The pathologist from the surgical specimen determines the pT and pN stages; the M stage is determined by imaging.
Tumor stage (pT)
- pT0 — No residual invasive tumor in the surgical specimen.
- pT1mi — Tumor 1 mm or smaller.
- pT1a — Tumor larger than 1 mm but 5 mm or smaller.
- pT1b — Tumor larger than 5 mm but 10 mm or smaller.
- pT1c — Tumor larger than 10 mm but 20 mm or smaller.
- pT2 — Tumor larger than 20 mm but 50 mm or smaller.
- pT3 — Tumor larger than 50 mm.
- pT4a — Tumor has grown into the chest wall.
- pT4b — Tumor has spread to the skin, causing ulceration or satellite nodules.
- pT4c — Both pT4a and pT4b.
- pT4d — Inflammatory breast cancer.
Nodal stage (pN)
- pN0 — No cancer in any lymph nodes examined.
- pN0(i+) — Isolated tumor cells only (≤0.2 mm) — not counted as positive.
- pN1mi — Micrometastases only (0.2–2 mm) in axillary lymph nodes.
- pN1a — Cancer in 1–3 axillary lymph nodes, with at least one deposit larger than 2 mm.
- pN1b — Cancer in ipsilateral internal mammary sentinel nodes (excluding isolated tumor cells).
- pN2a — Cancer in 4–9 axillary lymph nodes.
- pN2b — Cancer in internal mammary lymph nodes without axillary involvement.
- pN3a — Cancer in 10 or more axillary lymph nodes, or in infraclavicular lymph nodes.
- pN3b — Cancer in internal mammary and axillary lymph nodes.
- pN3c — Cancer in supraclavicular lymph nodes.
What is the prognosis for invasive lobular carcinoma?
The prognosis for invasive lobular carcinoma is generally favorable in the early stages, reflecting the fact that most cases are low grade, hormone receptor-positive, and diagnosed at a localized or regionally limited stage. Five-year and ten-year survival rates for hormone receptor-positive, early-stage ILC are excellent, often comparable to, or better than, those for invasive ductal carcinoma of similar stage and grade.
However, invasive lobular carcinoma has several distinctive prognostic features that distinguish it from invasive ductal carcinoma:
- Higher risk of late recurrence — Some studies show that ILC has a higher risk of distant recurrence beyond 5 years compared to invasive ductal carcinoma. Patients benefit from extended hormone therapy (up to 10 years) partly for this reason.
- Distinct metastatic pattern — When invasive lobular carcinoma does spread, it has a unique predilection for sites that differ from invasive ductal carcinoma. ILC commonly spreads to the bone, gastrointestinal tract (including the stomach and colon), ovaries, and peritoneum, rather than to the lungs, which are a more typical metastatic site for other breast cancers. This unique pattern means that patients may need tailored surveillance and that gastrointestinal or gynecologic symptoms should be investigated promptly.
- Subtype differences — The pleomorphic and solid subtypes are associated with a worse prognosis than the classic type. Grade 3 tumors and those with lymph node involvement carry a higher risk than grade 1 tumors without nodal disease.
- Excellent response to hormone therapy — The near-universal hormone receptor positivity of ILC means that most patients respond well to long-term hormone-blocking therapy, which substantially reduces recurrence risk.
Questions to ask your doctor
Your pathology report contains important information that will guide your care. The following questions may help you prepare for your next appointment.
- What subtype of invasive lobular carcinoma do I have — classic or pleomorphic — and does that affect my prognosis?
- What was the tumor size and Nottingham grade?
- What is the pathologic stage of my cancer (pT and pN)?
- Were any lymph nodes involved, and if so, how many?
- Were the surgical margins clear? Was the tumor completely removed?
- Was lymphovascular invasion present?
- Was E-cadherin loss confirmed on immunohistochemistry?
- What are my hormone receptor (ER and PR) results, and which hormone-blocking therapy do you recommend?
- What is my HER2 status — is my tumor HER2-negative, HER2-low, or HER2-positive?
- Will genomic testing (such as Oncotype DX) be performed, and is it reliable for invasive lobular carcinoma?
- Should I be referred for genetic counseling to assess for a CDH1 gene mutation or other inherited risk factors?
- If I received neoadjuvant therapy, what was my residual cancer burden score?
- Given ILC’s tendency to spread to the stomach, colon, and ovaries, what surveillance imaging or symptoms should I watch for?
- What additional treatment is recommended — radiation, chemotherapy, hormone therapy, or targeted therapy?
- How long will I need to take hormone therapy?


