by Jason Wasserman MD PhD FRCPC
February 6, 2026
Follicular thyroid carcinoma is a type of thyroid cancer that starts from follicular cells, the cells in the thyroid gland that produce thyroid hormones. The thyroid is a small, butterfly-shaped gland located at the front of the neck that helps regulate how the body uses energy.
This cancer is considered well differentiated, meaning the tumour cells still resemble normal thyroid cells under the microscope. What makes follicular thyroid carcinoma a cancer, rather than a benign tumour, is invasion. Invasion means the tumour cells have grown through the tumour capsule (the thin layer of tissue surrounding the tumour) and/or have entered nearby blood vessels.
This article explains how follicular thyroid carcinoma is diagnosed, classified, and evaluated, what features pathologists look for, and how these findings relate to prognosis and treatment.
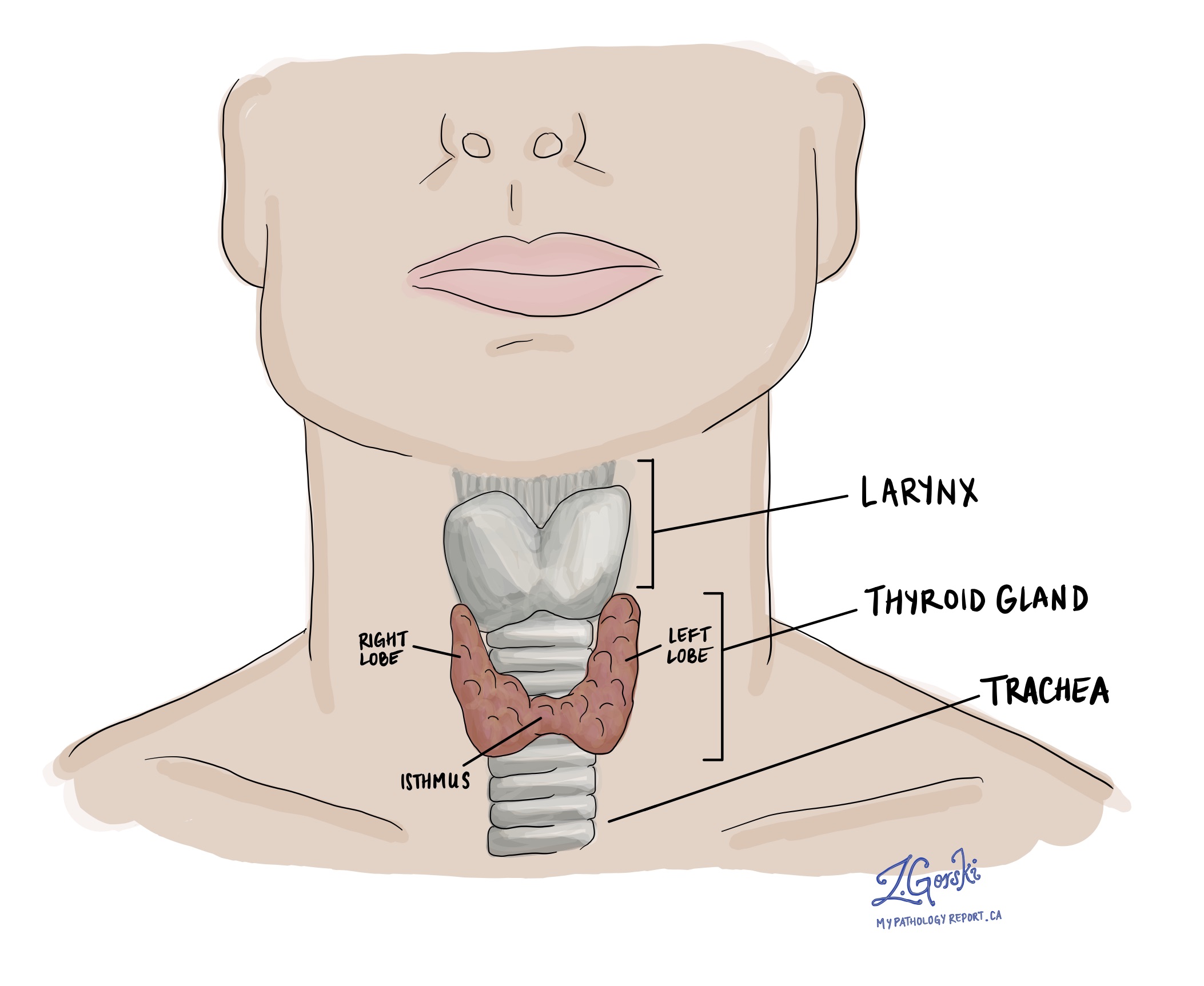
How common is follicular thyroid carcinoma?
Follicular thyroid carcinoma accounts for approximately 5–15% of thyroid cancers. It is less common than papillary thyroid carcinoma and occurs more often in adults than in children. Rates are higher in parts of the world where dietary iodine intake is low.
What are the symptoms of follicular thyroid carcinoma?
Many people first notice a painless lump in the neck, usually in the area of the thyroid gland. Some tumours are found incidentally during imaging or a routine physical examination.
Larger tumours may cause symptoms related to pressure on nearby structures, such as difficulty swallowing, hoarseness, or a feeling of tightness in the neck. Most patients have normal thyroid hormone levels, so symptoms of overactive or underactive thyroid are uncommon.
In a small percentage of cases, the cancer has already spread to distant organs, such as the lungs or bones. When this happens, symptoms may come from the site of spread, such as bone pain or shortness of breath.
What causes follicular thyroid carcinoma?
The exact cause is not always known. Several factors are associated with an increased risk, including iodine deficiency, radiation exposure (especially during childhood), and inherited tumour syndromes such as PTEN hamartoma tumour syndrome (Cowden syndrome), DICER1 syndrome, Werner syndrome, and Carney complex.
Most cases are sporadic, meaning they occur by chance and are not inherited.
How is follicular thyroid carcinoma diagnosed?
Diagnosing follicular thyroid carcinoma requires several steps because this tumour cannot be definitively diagnosed on imaging or needle biopsy alone.
The diagnostic process usually includes imaging studies, fine-needle aspiration (FNA) biopsy, surgical removal of the tumour, and careful microscopic examination. The final diagnosis is almost always made after surgery, when the tumour capsule and nearby blood vessels can be fully examined.
Imaging
Ultrasound is typically the first imaging test. Follicular thyroid carcinoma often appears as a solid, well-defined thyroid nodule, sometimes with a surrounding halo caused by the tumour capsule. Imaging cannot reliably distinguish follicular thyroid carcinoma from a benign follicular adenoma.
Fine-needle aspiration (FNA)
FNA can show that a tumour is made of follicular cells, but it cannot determine whether the tumour is benign or malignant. This is because invasion cannot be assessed on small biopsy samples. As a result, FNA reports often use terms such as follicular neoplasm or suspicious for follicular neoplasm, and surgery is required to establish the diagnosis.
Microscopic features
Under the microscope, follicular thyroid carcinoma is made up of follicular cells arranged in follicles (round spaces that normally store thyroid hormone) or in solid or trabecular growth patterns. The tumour cells resemble those in a benign follicular adenoma, but the key difference is their invasive nature.
Capsular invasion means the tumour cells have completely grown through the tumour capsule. Vascular invasion means tumour cells are found inside blood vessels, often attached to the vessel wall or mixed with a blood clot. Identifying at least one focus of capsular or vascular invasion is required to make the diagnosis of follicular thyroid carcinoma.
Immunohistochemistry
Immunohistochemistry is a laboratory test that uses antibodies to detect specific proteins in tumour cells. In follicular thyroid carcinoma, tumour cells typically express thyroglobulin, TTF-1, and PAX8, confirming that the tumour started from thyroid follicular cells.
These tests help confirm tumour origin and rule out other types of cancer. However, immunohistochemistry cannot reliably distinguish follicular thyroid carcinoma from follicular adenoma. The diagnosis still depends on identifying invasion under the microscope.
Tumour classification (subtypes)
Follicular thyroid carcinoma is divided into subtypes based on the extent of tumour invasion of surrounding structures. This classification is important because it strongly influences prognosis and follow-up.
Minimally invasive follicular thyroid carcinoma
Minimally invasive tumours show capsular invasion only, meaning tumour cells have grown through the capsule but have not entered blood vessels. These tumours are usually well contained and behave in an indolent manner. When completely removed, the prognosis is excellent, and additional treatment is often not required.
Encapsulated angioinvasive follicular thyroid carcinoma
A capsule surrounds these tumours, but they show vascular invasion, meaning tumour cells are found inside blood vessels. Blood vessels act like highways, carrying tumour cells to distant organs.
Pathologists further divide vascular invasion into:
-
Limited vascular invasion: tumour cells are found in fewer than 4 blood vessels.
-
Extensive vascular invasion: tumour cells are found in 4 or more blood vessels.
Tumours with limited vascular invasion have a good-to-intermediate prognosis, whereas those with extensive vascular invasion carry a higher risk of distant spread and require closer follow-up.
Widely invasive follicular thyroid carcinoma
Widely invasive tumours show extensive infiltration into the surrounding thyroid tissue or nearby soft tissues. These tumours do not always have a complete capsule, making invasion more obvious. Widely invasive tumours often involve multiple blood vessels and have the highest risk of recurrence and distant metastasis.
Lymph nodes
Lymph nodes are small immune organs that filter lymphatic fluid. Cancer cells can spread from the thyroid to nearby lymph nodes through lymphatic vessels.
Follicular thyroid carcinoma spreads to lymph nodes less often than papillary thyroid carcinoma, but it can still occur. Any lymph nodes removed during surgery are examined under the microscope and reported as positive or negative for tumour cells.
Finding tumour cells in lymph nodes may increase the cancer stage and influence recommendations for additional treatment and follow-up.
Margins
A margin is the edge of tissue removed during surgery. Pathologists examine margins to determine whether tumour cells are present at the cut edge of the tissue.
A negative margin means no tumour cells are seen at the edge, suggesting the tumour was completely removed. A positive margin means tumour cells are present at the edge, indicating that some tumour may remain in the body. Margin status helps guide decisions about additional treatment and surveillance.
Pathologic stage (pTNM)
The pathologic stage for follicular thyroid carcinoma is based on the TNM staging system, which considers the primary tumour (T), lymph nodes (N), and distant metastasis (M). In general, a higher stage indicates more advanced disease and a worse prognosis.
Tumour stage (pT)
The tumour stage is based on tumour size and whether tumour cells extend beyond the thyroid:
-
T1: tumour ≤ 2 cm, confined to the thyroid.
-
T2: tumour > 2 cm but ≤ 4 cm, confined to the thyroid.
-
T3: tumour > 4 cm or minimal extension into nearby neck muscles.
-
T4: tumour extends into major structures such as the trachea, larynx, or esophagus.
Nodal stage (pN)
-
N0: no tumour cells found in lymph nodes.
-
N1: tumour cells found in lymph nodes.
-
NX: No lymph nodes submitted for examination.
Biomarkers for thyroid cancer
Biomarkers are tests performed on tumour tissue to detect changes in genes or proteins that help explain how the cancer developed and how it may behave. In thyroid cancer, biomarker testing is used to support the diagnosis, estimate the risk of recurrence, and identify patients who may benefit from targeted treatments. Not every thyroid cancer requires biomarker testing, and the specific genes tested depend on the tumour type and clinical situation.
BRAF
The BRAF gene helps control normal cell growth by sending signals that tell cells when to divide. In thyroid cancer, a specific change called BRAF V600E permanently switches on the growth signal, allowing tumour cells to grow and survive when they should not. This mutation is most commonly seen in papillary thyroid carcinoma and is associated with a higher risk of recurrence in some patients, especially when found with other high-risk features. Tumours with BRAF mutations may respond to targeted medications called BRAF inhibitors, sometimes combined with MEK inhibitors, particularly in advanced or radioactive iodine–resistant disease.
RAS (NRAS, HRAS, KRAS)
The RAS family of genes plays an important role in transmitting growth signals inside normal thyroid cells. When a RAS gene is mutated, the signals it encodes become overactive, promoting cell growth and survival. RAS mutations are common in follicular-patterned thyroid tumours, including follicular thyroid carcinoma, oncocytic carcinoma, and NIFTP. While RAS mutations are not specific for cancer and can be seen in benign tumours, their presence helps explain tumour development and may influence how closely a tumour is followed. At present, there are no standard RAS-targeted therapies for thyroid cancer, but these tumours may respond differently to certain systemic treatments.
TERT promoter
The TERT promoter controls telomerase activity, a protein that helps cells maintain their chromosomes during repeated divisions. In normal cells, telomerase activity is tightly controlled, but mutations in the TERT promoter allow tumour cells to divide indefinitely. In thyroid cancer, TERT promoter mutations are associated with more aggressive behaviour, a higher risk of recurrence, and worse prognosis, particularly when combined with BRAF or RAS mutations. While there is no specific TERT-targeted therapy, the presence of this mutation often influences treatment intensity and follow-up planning.
RET
The RET gene produces a receptor involved in cell growth and development. In thyroid cancer, RET can contribute to cancer in two different ways: through gene rearrangements (most often in papillary thyroid carcinoma) or point mutations (characteristic of medullary thyroid carcinoma). These changes lead to continuous growth signalling. RET alterations are important because highly effective RET-targeted therapies are available and can be used in advanced or metastatic disease, often with fewer side effects than traditional chemotherapy.
NTRK (NTRK1, NTRK3)
The NTRK genes normally help nerve cells grow and communicate. In some thyroid cancers, NTRK genes become abnormally fused to other genes, creating a protein that continuously drives tumour growth. Although uncommon, identifying an NTRK fusion is very important because tumours with this change often respond dramatically to TRK inhibitor therapies, which are highly effective even in advanced disease.
PPARG (PAX8::PPARG rearrangement)
The PPARG gene regulates cell metabolism and differentiation. In some follicular thyroid carcinomas, PPARG becomes abnormally fused to another gene, PAX8, creating a fusion that disrupts normal growth control. Tumours with this rearrangement are often well differentiated and may behave less aggressively, although exceptions exist. There are currently no standard targeted treatments specifically for PPARG rearrangements, but this result helps clarify tumour type and expected behaviour.
PIK3CA and AKT1
The PIK3CA and AKT1 genes are part of a pathway that controls cell growth, metabolism, and survival. Mutations in these genes can make tumour cells more resistant to normal growth controls. In thyroid cancer, these mutations are more often seen in advanced, poorly differentiated, or high-grade tumours. Their presence may indicate a higher-risk cancer and, in some cases, eligibility for targeted therapies that affect the PI3K/AKT/mTOR pathway, usually in clinical trials or advanced settings.
TP53
The TP53 gene is often called the “guardian of the genome” because it helps prevent damaged cells from dividing. When TP53 is mutated, this protective mechanism is lost, allowing cells with genetic damage to survive and multiply. TP53 mutations are uncommon in well-differentiated thyroid cancers but are frequently seen in poorly differentiated and anaplastic thyroid carcinoma. When identified, a TP53 mutation suggests a more aggressive tumour and may influence treatment decisions and prognosis, although there is no direct TP53-targeted therapy at present.
CTNNB1 (β-catenin)
The CTNNB1 gene produces β-catenin, a protein involved in cell adhesion and growth signalling. Abnormal activation of this pathway can lead to uncontrolled cell growth. CTNNB1 mutations are rare in typical differentiated thyroid cancers but are more often found in high-grade or poorly differentiated tumours. Their presence supports a diagnosis of aggressive disease and helps explain resistance to standard treatments.
ALK
The ALK gene normally plays a role in early cell development. In rare thyroid cancers, ALK becomes abnormally fused to another gene, leading to constant growth signalling. Although uncommon, identifying an ALK rearrangement is important because ALK-targeted therapies may be effective in patients with advanced disease.
Prognosis
The prognosis for follicular thyroid carcinoma depends mainly on the extent of invasion. Minimally invasive tumours have an excellent outcome. Tumours with limited vascular invasion have a slightly higher risk but still do well with appropriate treatment. Widely invasive tumours and those with extensive vascular invasion carry a higher risk of recurrence and distant spread.
The presence of distant metastases and certain molecular changes, such as TERT promoter mutations, is associated with worse outcomes.
Questions to ask your doctor
-
What subtype of follicular thyroid carcinoma do I have?
-
Was capsular or vascular invasion present?
-
How extensive was the vascular invasion?
-
Were any lymph nodes involved?
-
Will I need radioactive iodine therapy?
-
What is my long-term risk of recurrence?
-
How often will I need follow-up tests or imaging?


